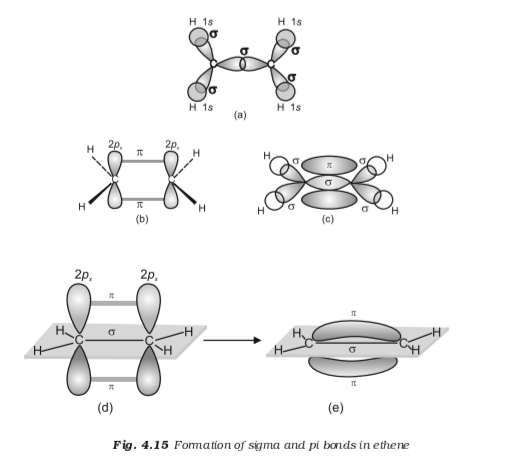
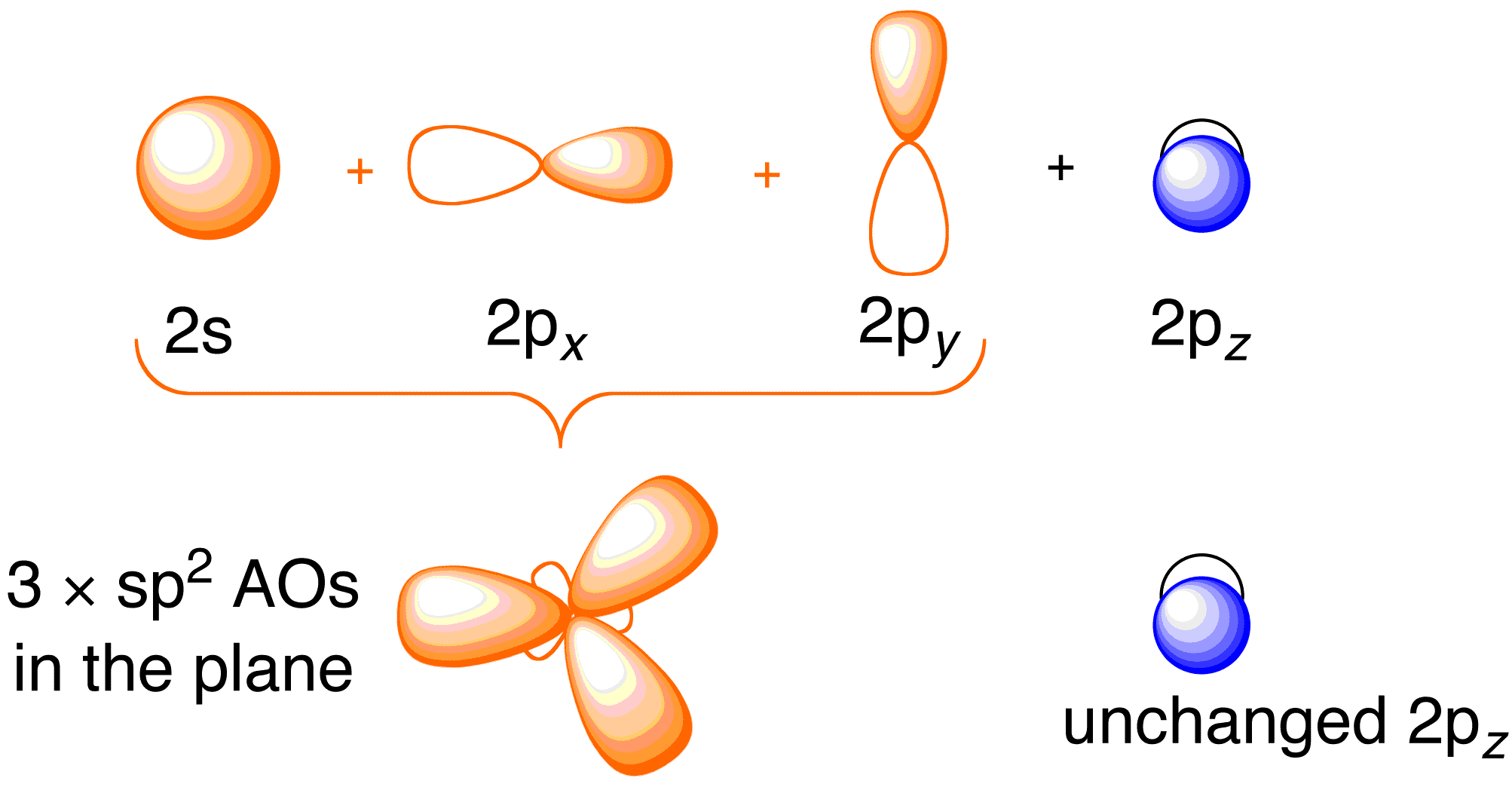Why didn't the 3rd P orbital of carbon in C2H4 take part in hybridization as it took part in Hybridization in CH4? - Quora
How to determine C2H4 hybridization when there are 0 lone pairs and 3 sigma bonds because that is 4 - Quora

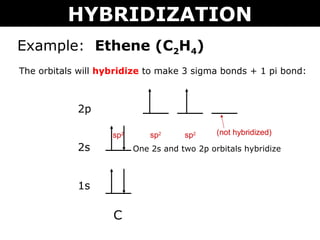
Hybridization|sp3,sp2,sp hybridization |Hybridization of carbon,nitrogen| c2h4,c2h2,nh3 hybridization | chemistry, nitrogen, carbon | Hybridization is the mixing up of atomic orbitals to form new orbitals. The orbital hybridization may be of different ...

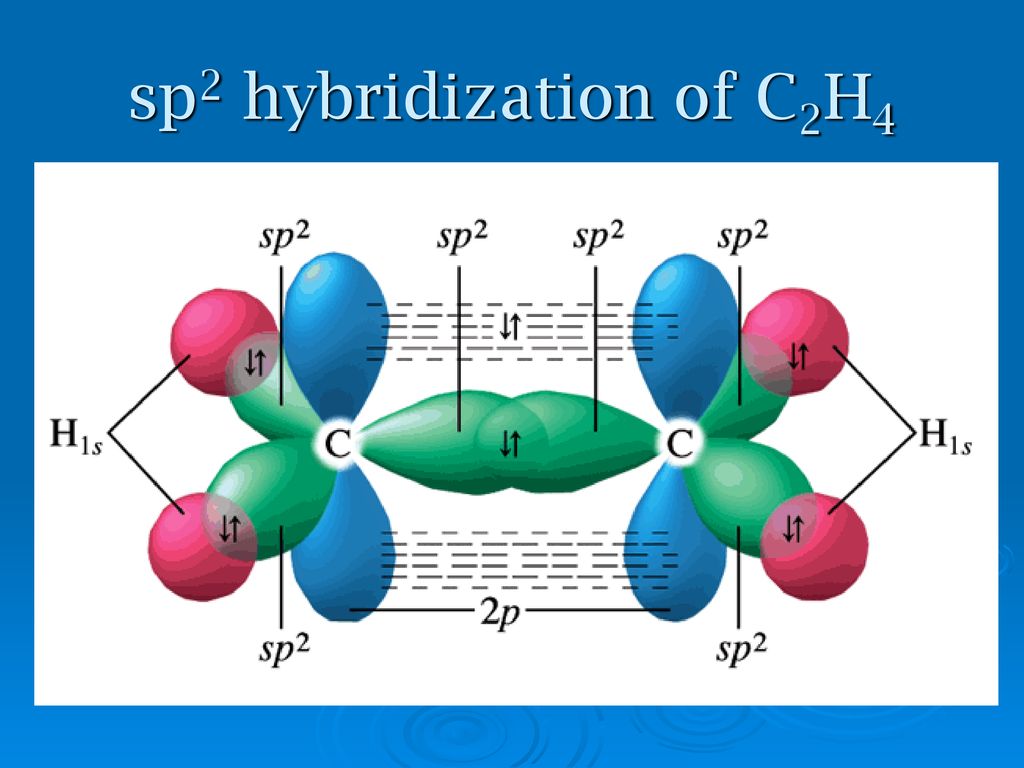
molecular orbital picture of ethylene, formation of sp2 hybridisation. 28282149 Vector Art at Vecteezy
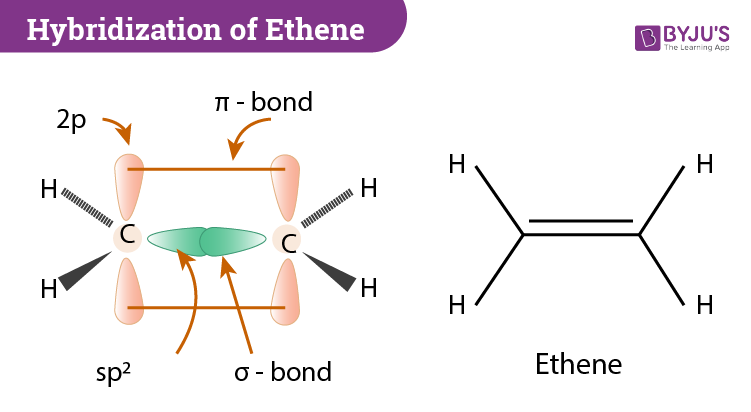
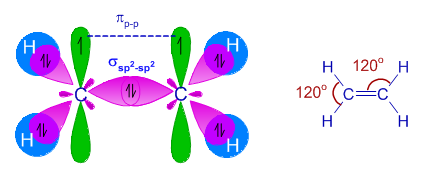
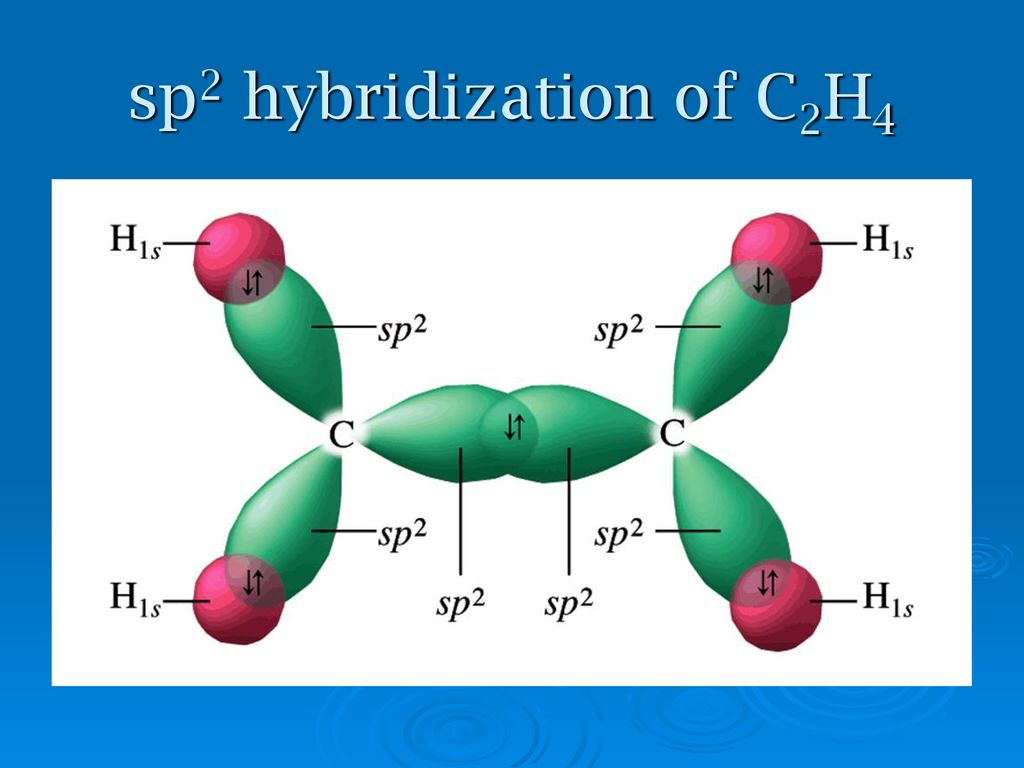
Describe briefly the structure of C2H4 on the basis of hybridisation. - Sarthaks eConnect | Largest Online Education Community

Explain what is meant by the term "hybridization" in molecular orbital theory and show how the concept can be used to explain the structure and bonding in ethane C2H6, ethene C2H4, and