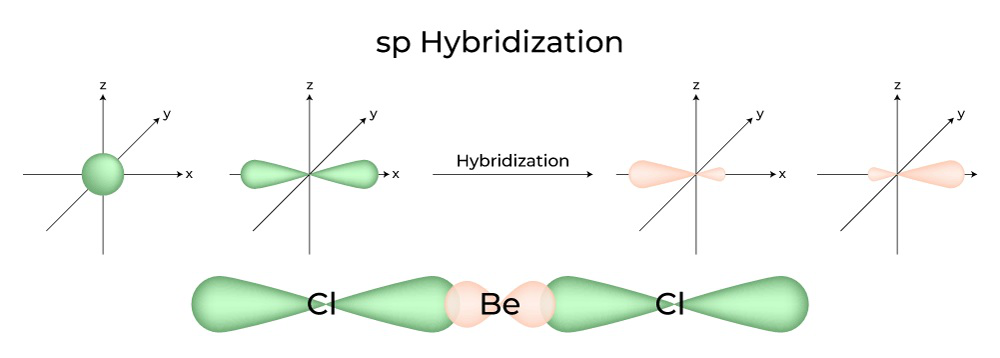
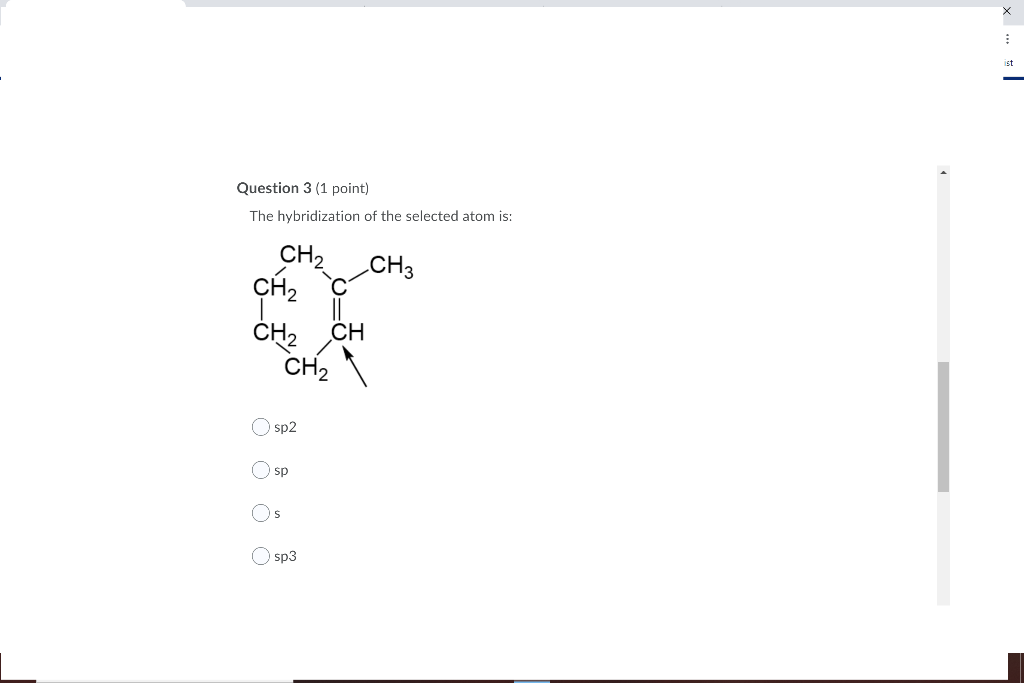
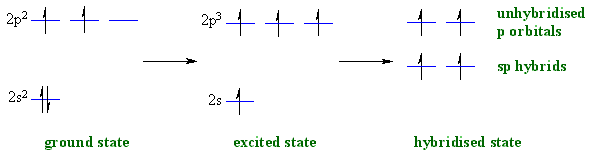
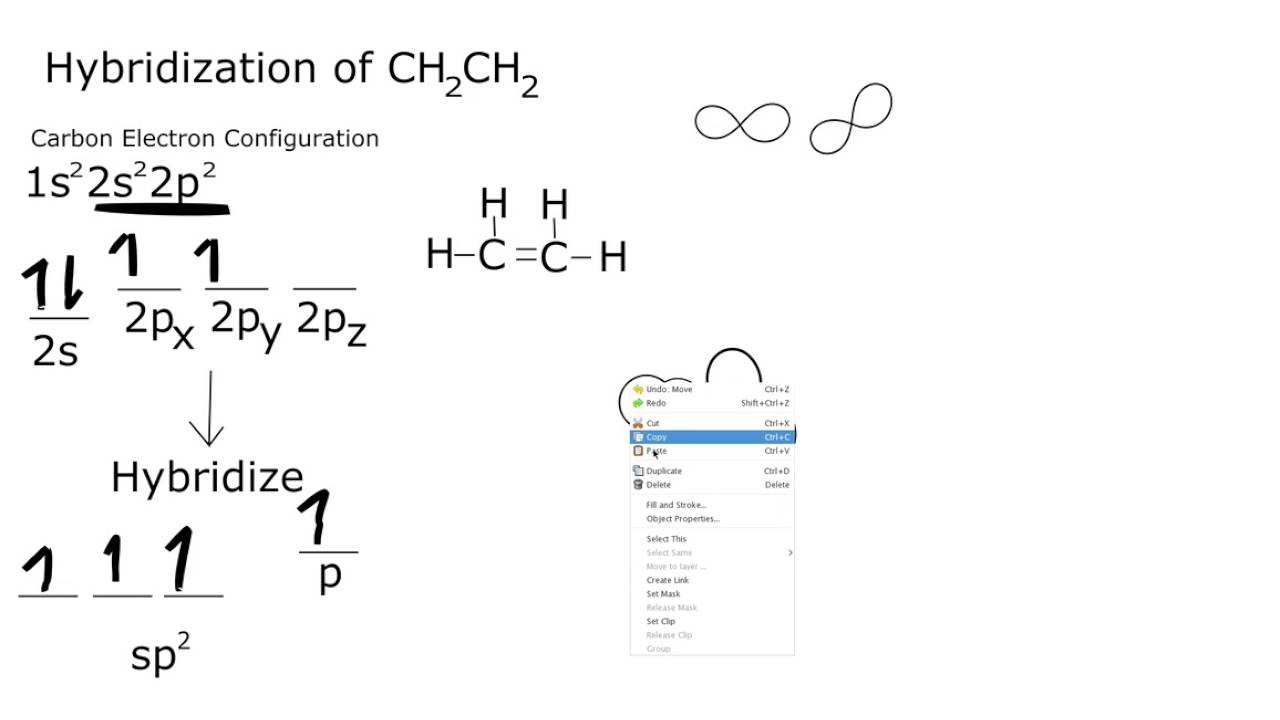
8.26 | For the molecule allene, H2 C = C = CH2, give the hybridization of each carbon atom. Will the - YouTube

Draw an orbital picture of allene, H2C=C=CH2. What hybridization must the central carbon atom have to form two double bonds? What shape does allene have? | Homework.Study.com
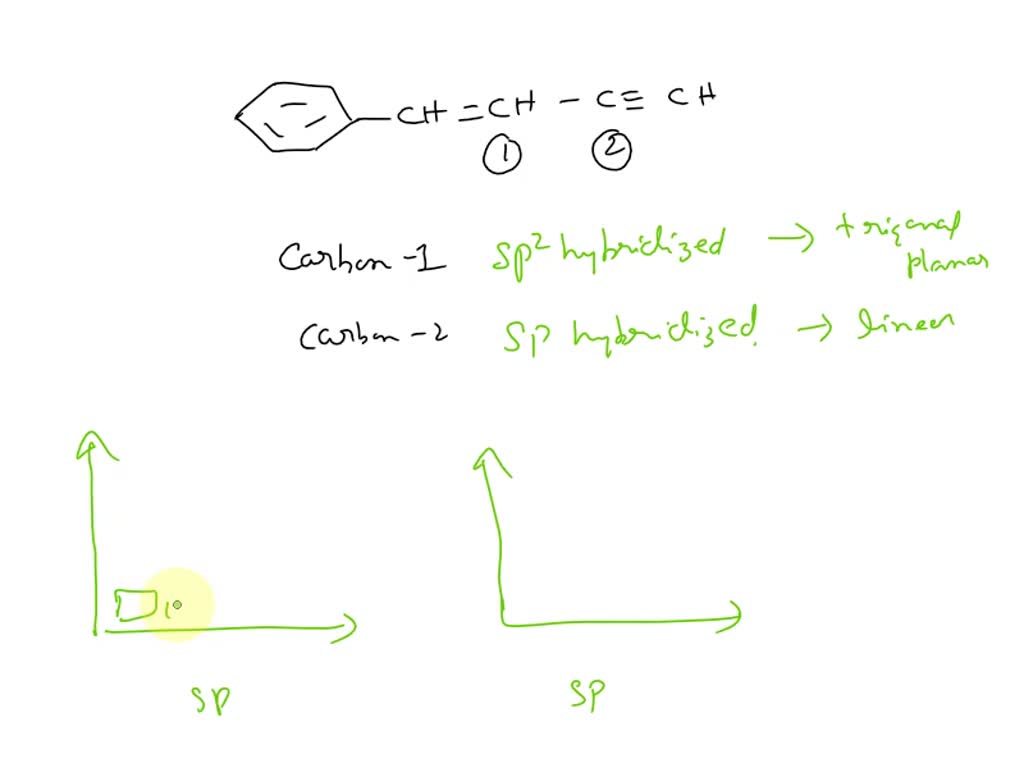
SOLVED: What is the hybridization of the carbon atoms numbered 1 and 2, respectively, in the following structure: CH=CH-C≡C-CH2?

Prepare a sketch of the molecule CH3CCl = CH2 showing orbital overlaps. Identify the type of hybridization of atomic orbitals on each carbon atom. | Homework.Study.com
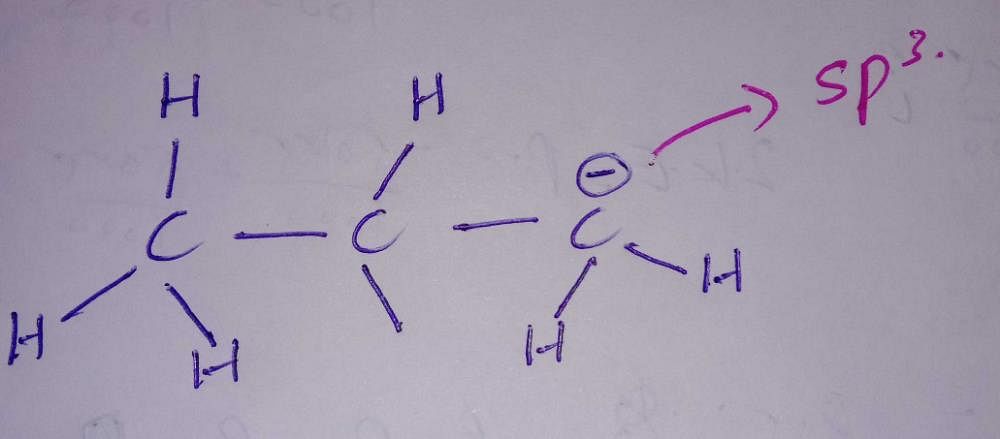
Hybridisation of the third carbon in CH3-CH2-CH2 (-) ( There's negative charge in third carbon) ? - EduRev NEET Question