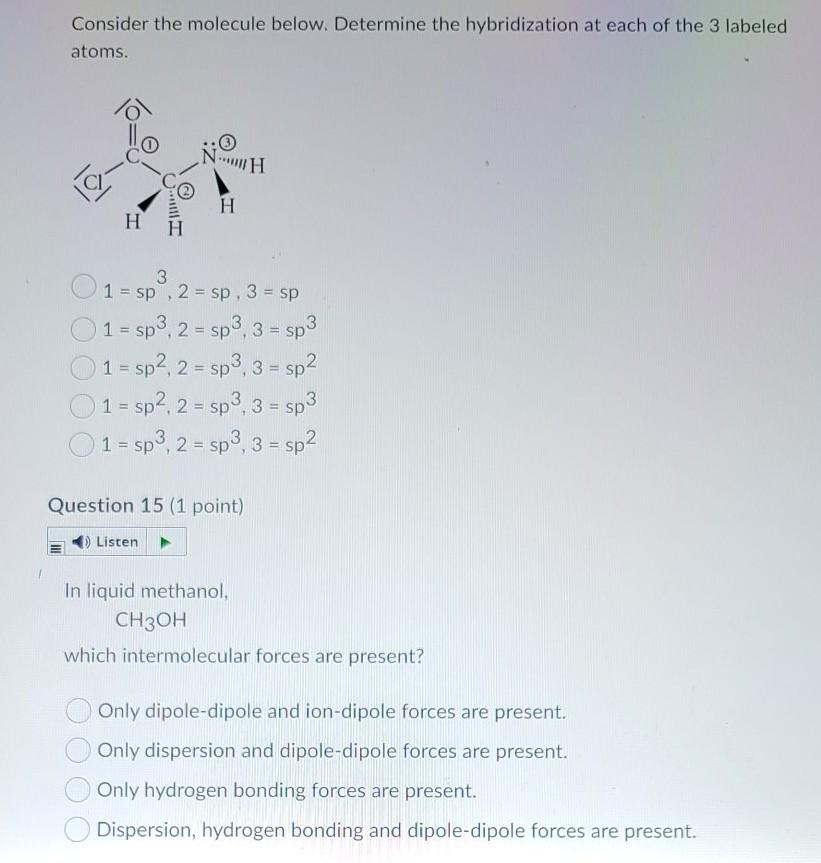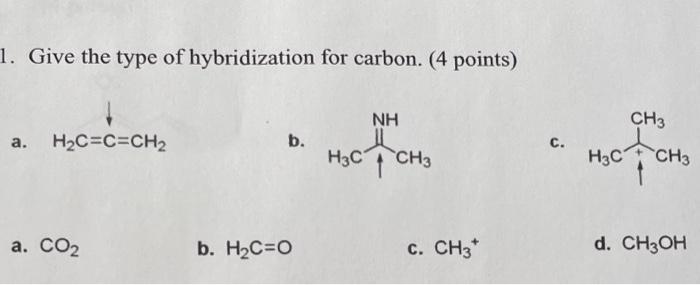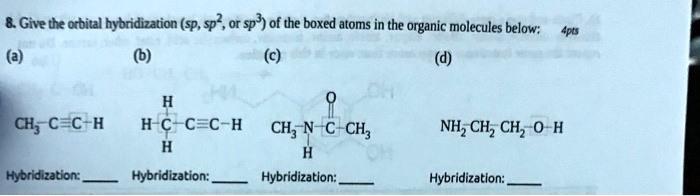
SOLVED: Give the orbital hybridization (SP, sp2, or sp3) of the boxed atoms in the organic molecules below: CH3: C-C-H H-C-C-C-H CH3: N-C-C-H NH3 CH2CH2 CH3OH Hybridization: sp3 Hybridization: sp3 Hybridization: sp3

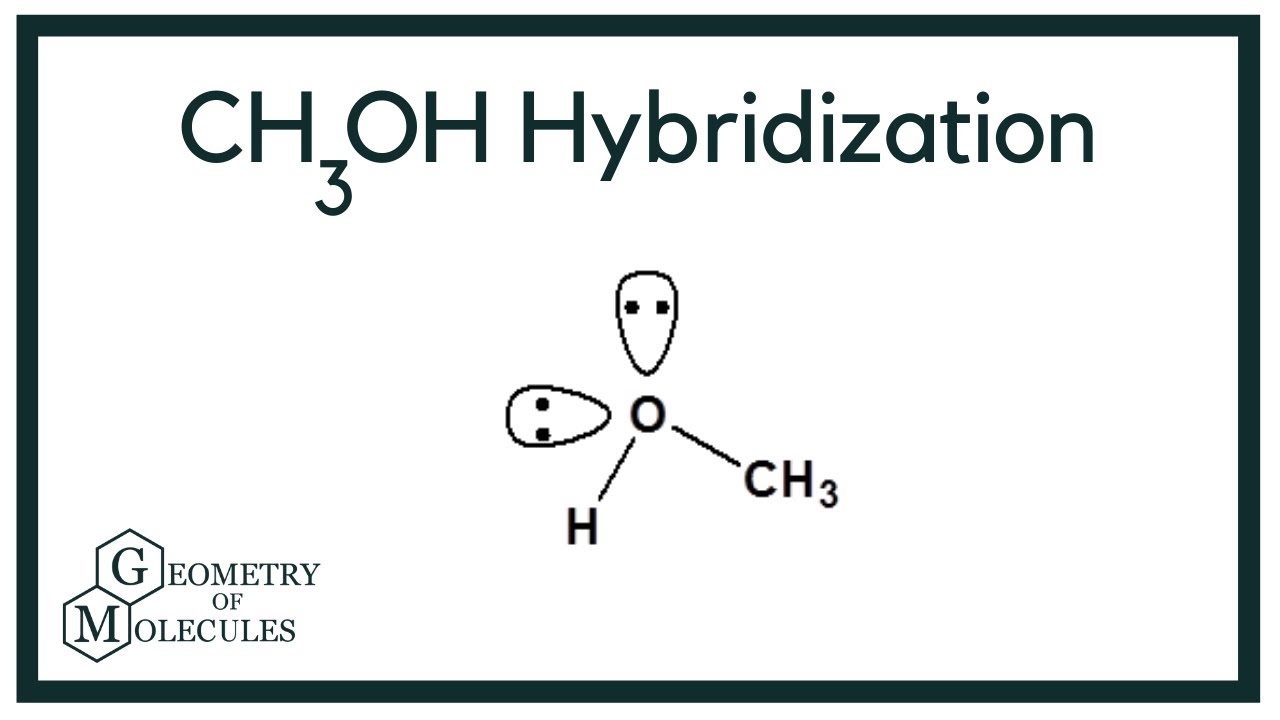
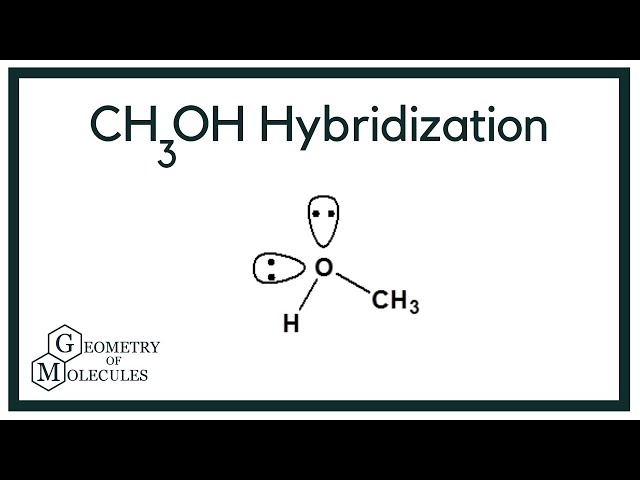
What orbitals are used to form each bond in methanol, ch3oh? be sure to answer all parts? - brainly.com

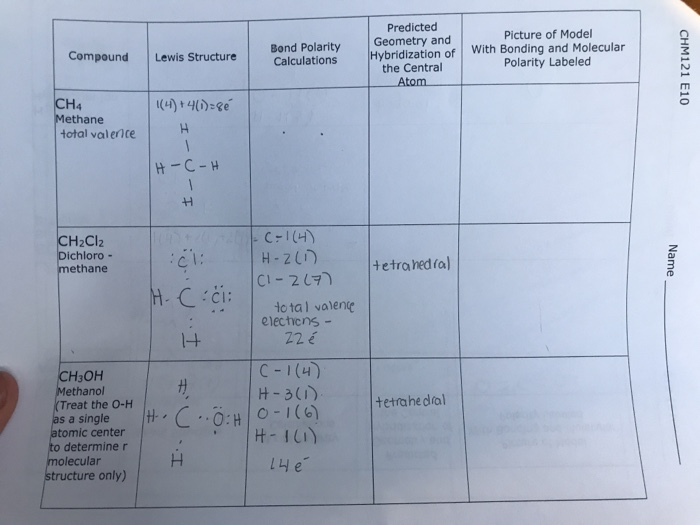
SOLVED: What is the hybrid orbital set (hybridization) used by each of the following molecules? H2O, NH3, CH4, C2H2, CH3OH. Specify the electron-pair and molecular geometry for each underlined atom in the

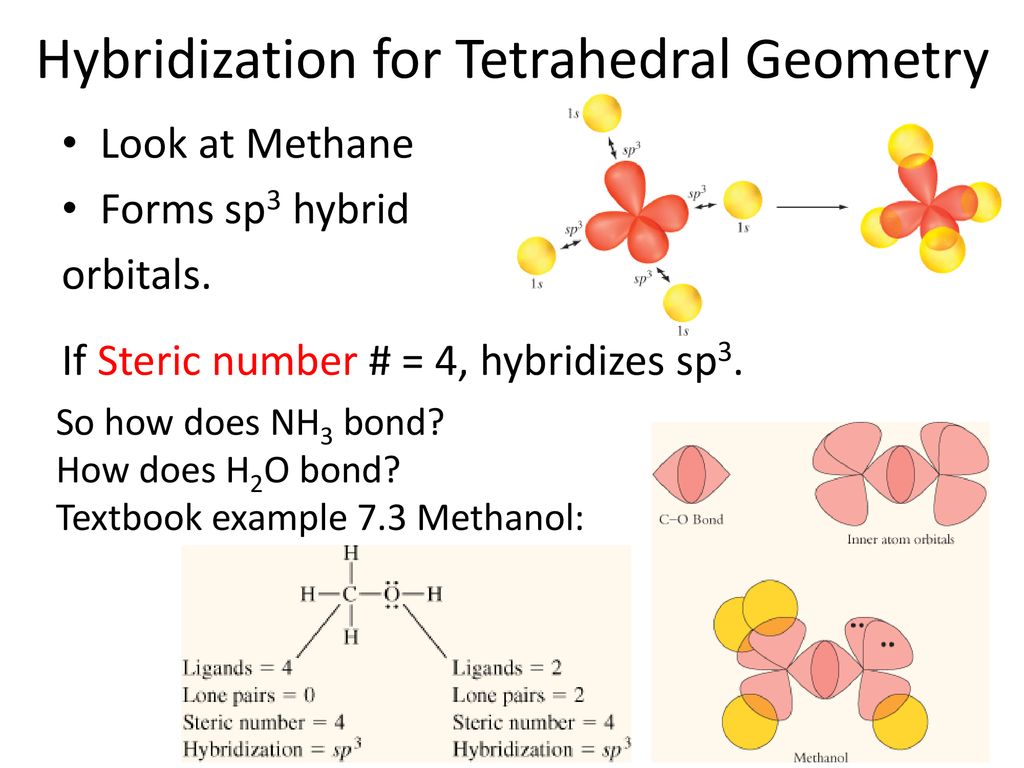
Methanol (CH3OH) Lewis structure, molecular geometry or shape, bond angle, hybridization | Molecular geometry, Molecular, Molecular shapes