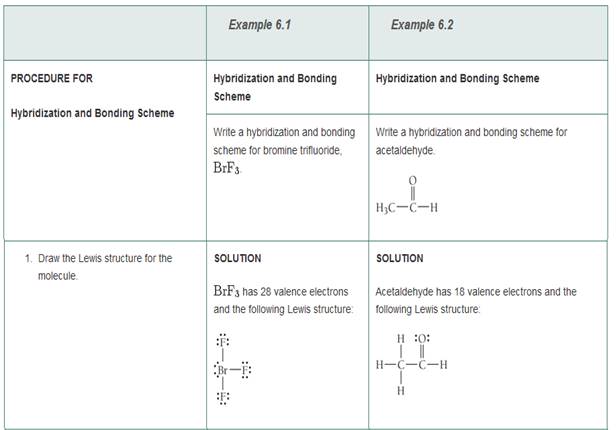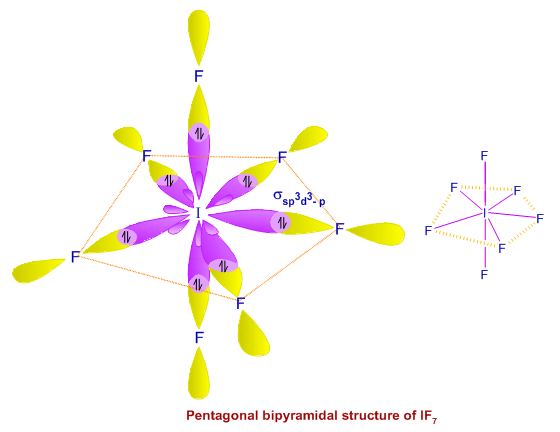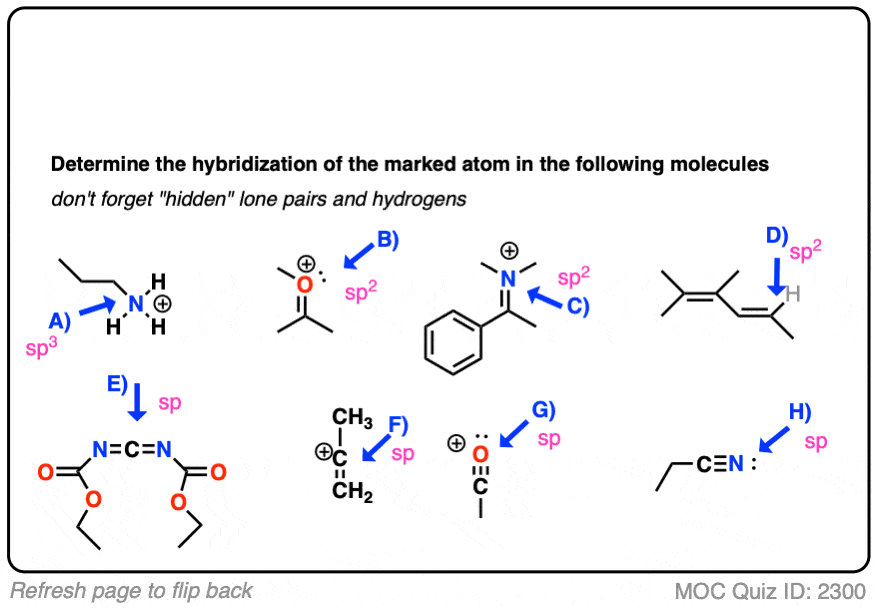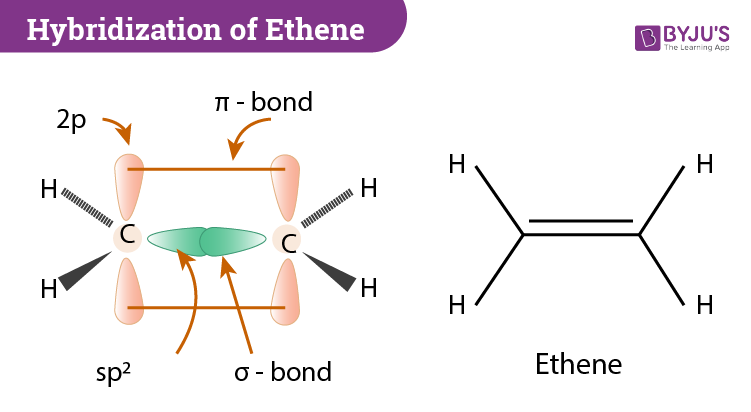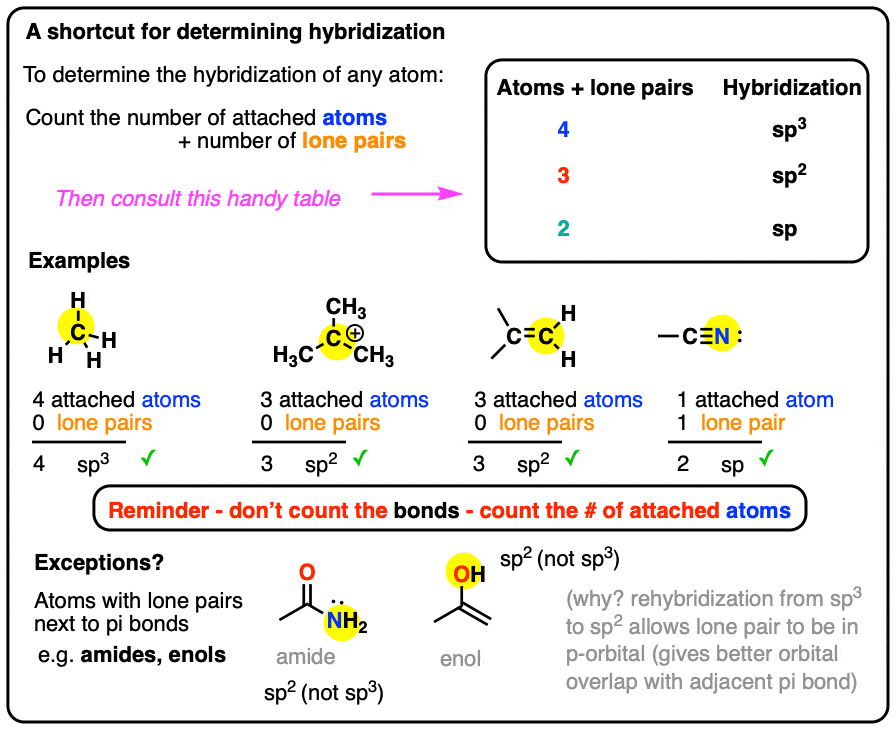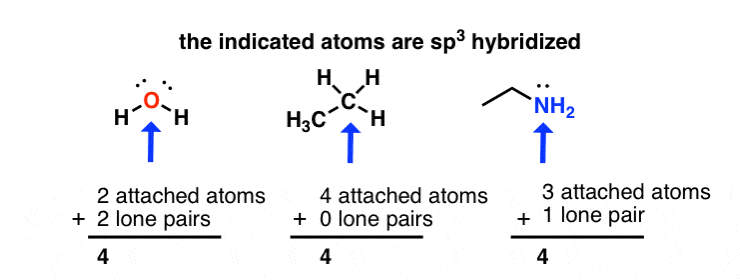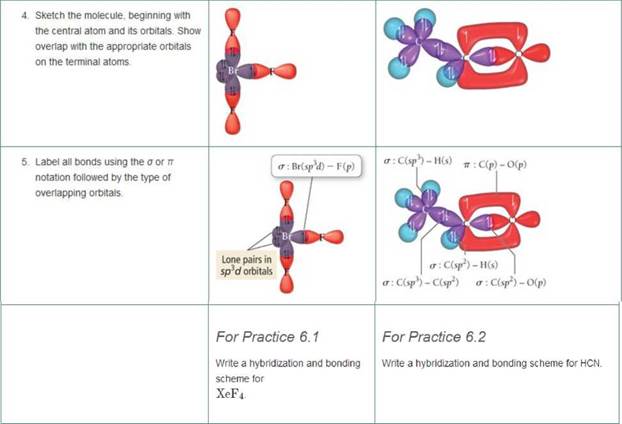
Write a hybridization and bonding scheme for each molecule. Sketch the molecule, including overlapping orbitals, and label all bonds using the notation shown in Examples 6.1 and 6.2 CCI 4 NH 3 OF 2 CO 2 | bartleby

Determining Hybridization of Valence Orbitals in Covalent Molecules using VSEPR Theory | Chemistry | Study.com

SOLVED: Determine the hybridization scheme for each of the four labeled atoms: 4 2 C 3 H 1=sp2, 2=sp?, 3=sp?, 4=sp 1=sp, 2=sp?, 3=sp?, 4=sp" 1=sp2, 2=sp?, 3=sp?, 4=sp 1=sp2, 2=sp?, 3=sp?,