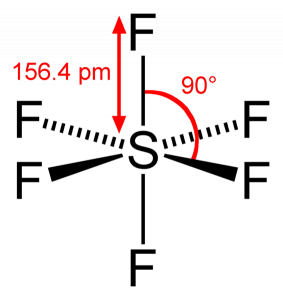
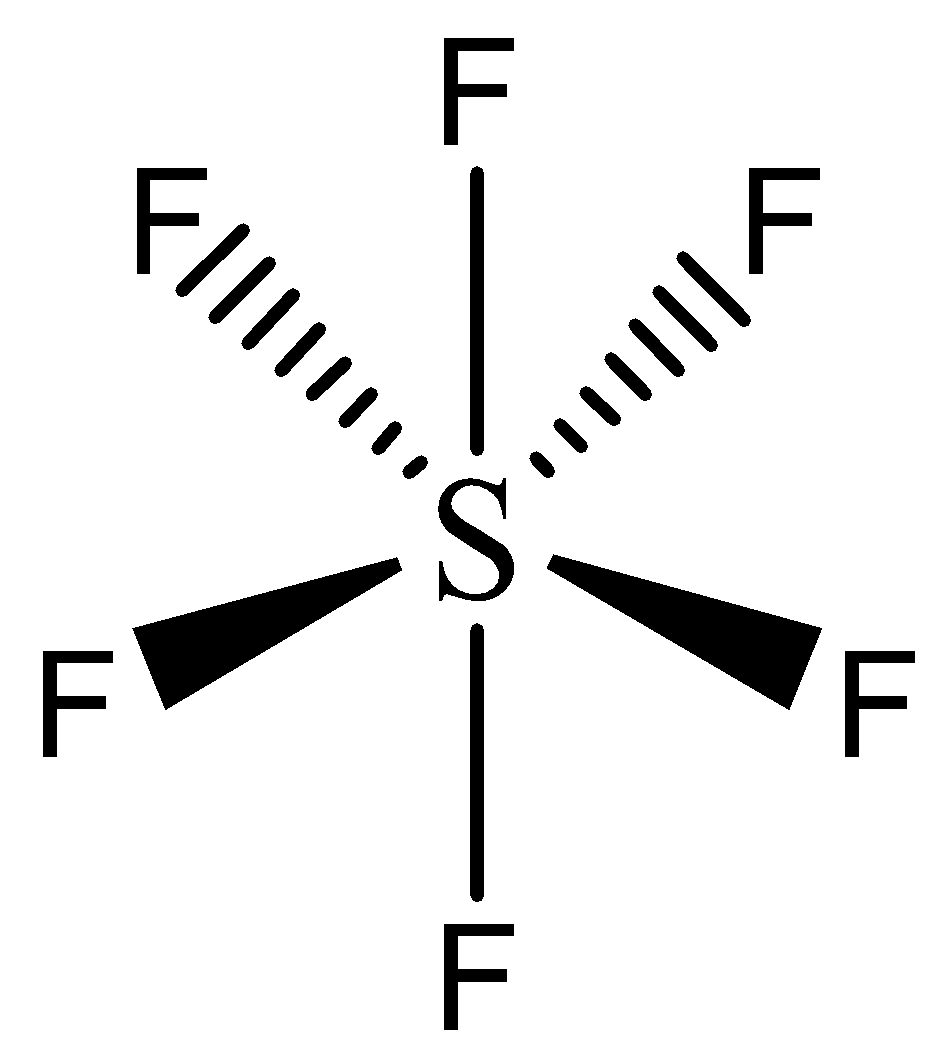
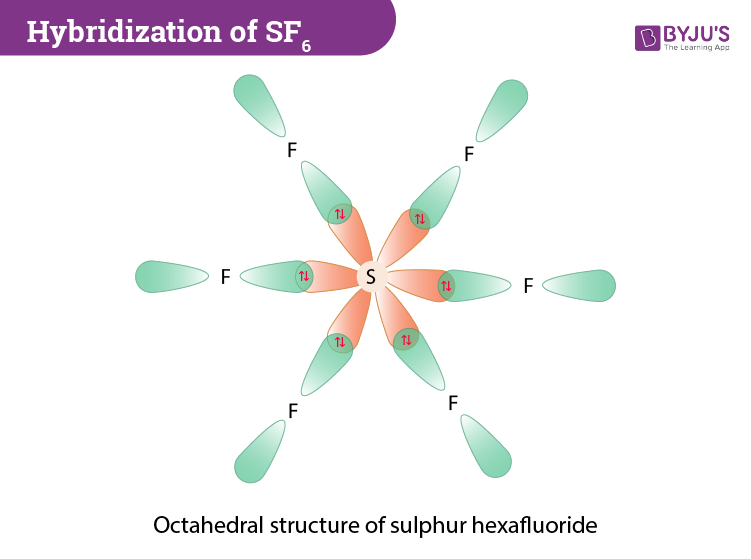
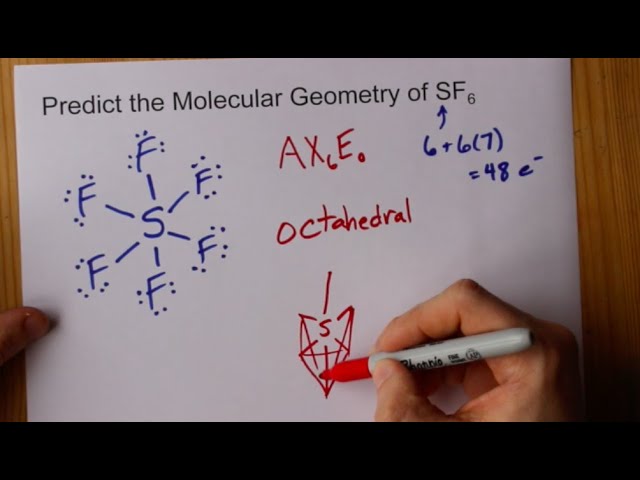
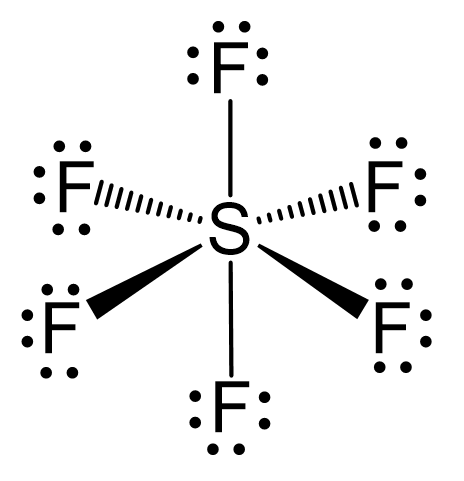
For the molecule SF6, determine its (a) electronic geometry, (b) number of nonbonding domains on the central atom, and (c) polarity. | Homework.Study.com

The sp3d2 Hybridization and Octahedral Geometry | Molecular geometry, Chemistry, Electron configuration
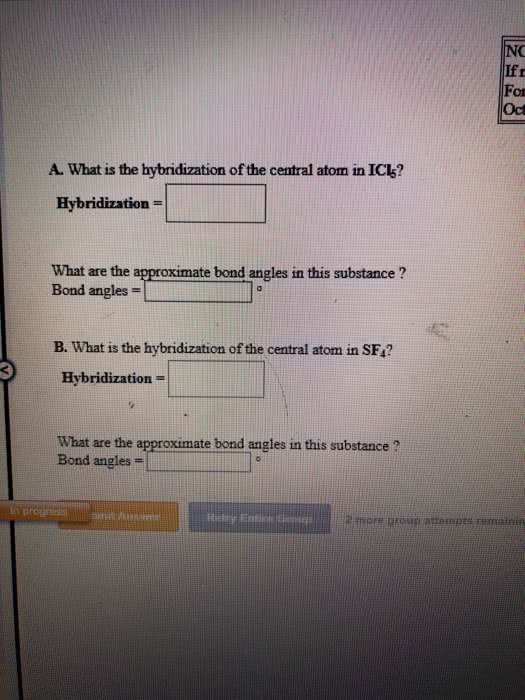
OneClass: NOTE If more For exa Octahe A. What is the hybridization of the central atom in SF6? Hybrid...
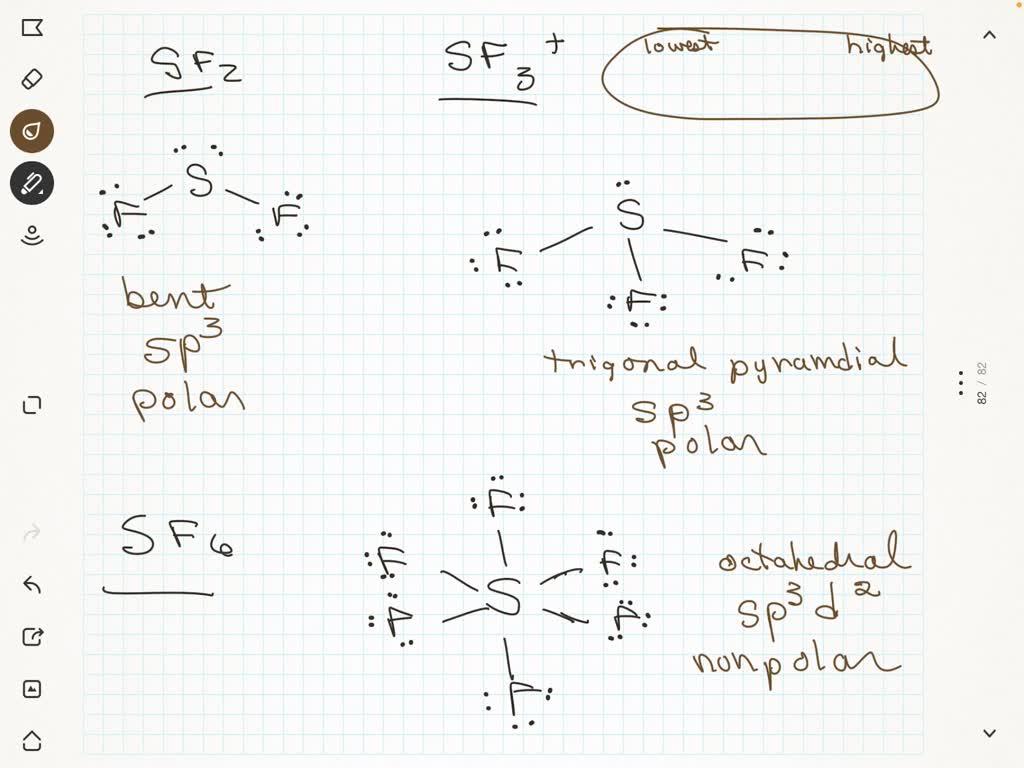
SOLVED: Consider the following molecules: SF2, SF3+, SF6. Draw the Lewis dot structures, determine VSEPR shapes, give hybridization of the central atoms, and predict whether the molecules are polar. Put in order

Chemistry - Molecular Structure (35 of 45) s-p3-d2 Hybridization - Sulfur Hexafloride - SF6 - YouTube

What atomic or hybrid orbitals make up the sigma bond between S and F in sulfur hexafluoride, SF6? | Homework.Study.com
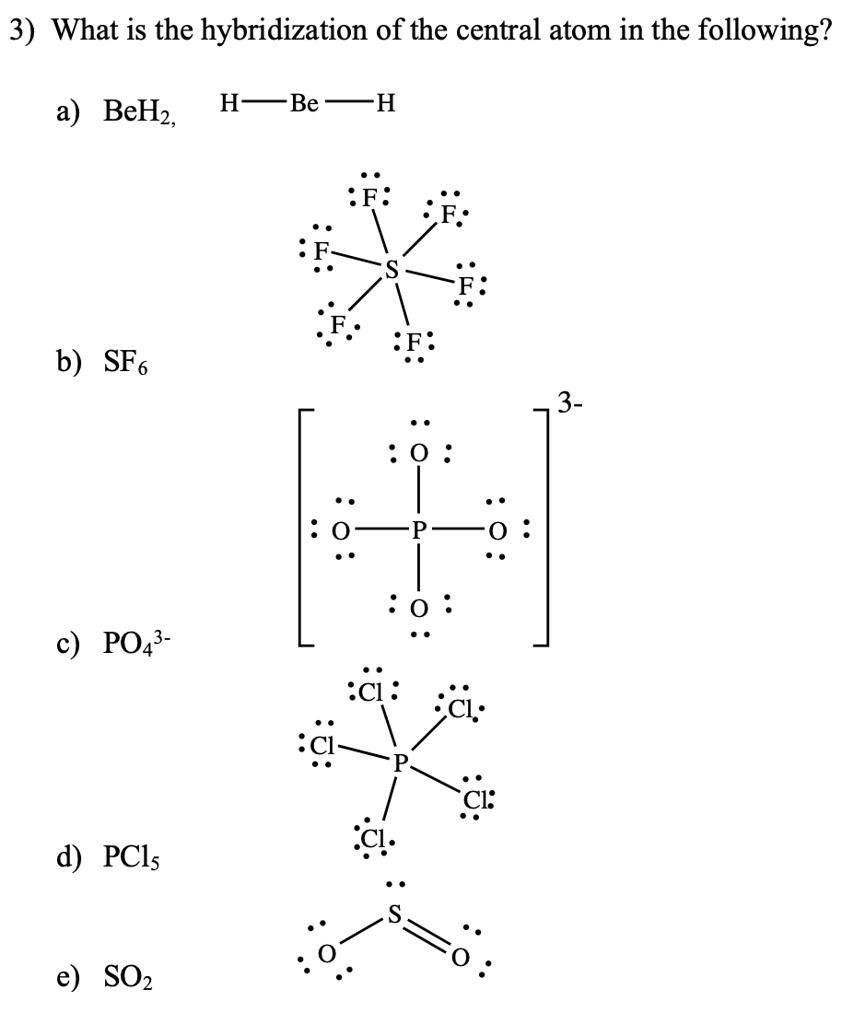
SOLVED: What is the hybridization of the central atom in the following? H2O a) BeH2, b) SF6 c) PO3^- d) PCl5 e) SO2

In which of the following the central atom is sp3 hybridised?a)NH4+ b)BF3c) SF6 d)PCl5Correct answer is option 'A'. Can you explain this answer? - EduRev NEET Question