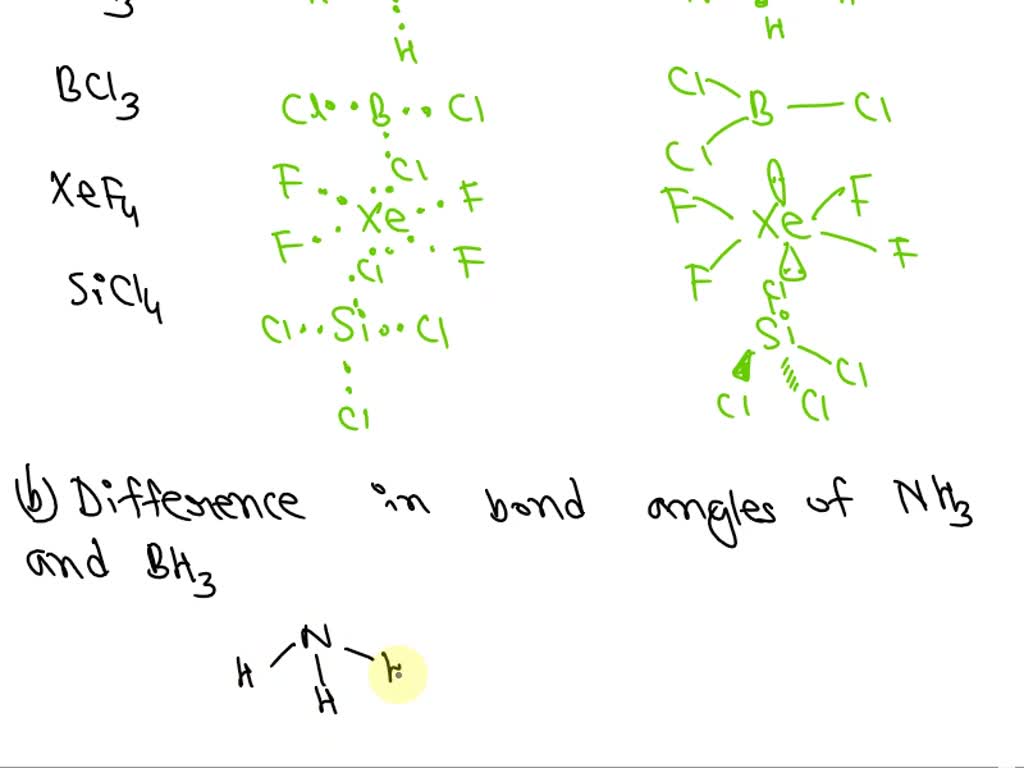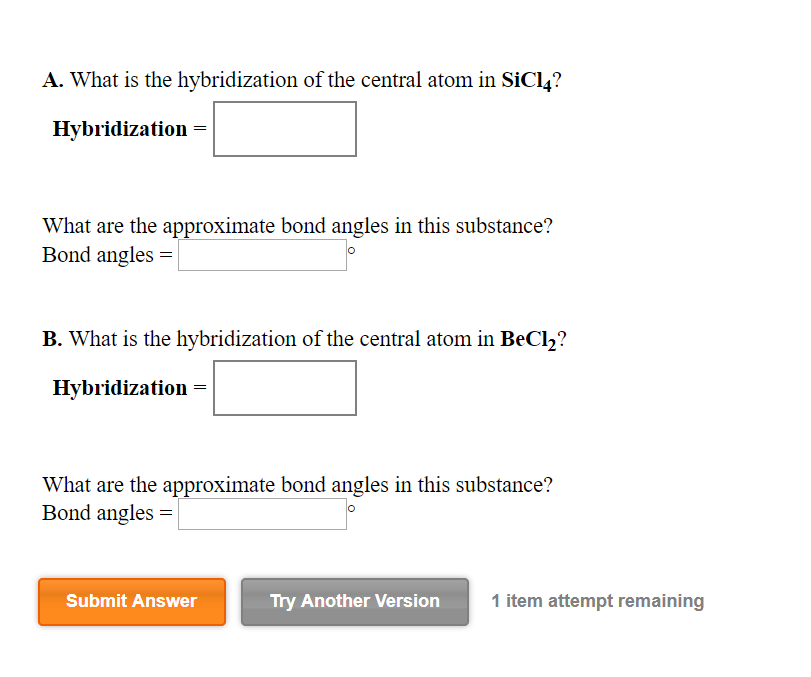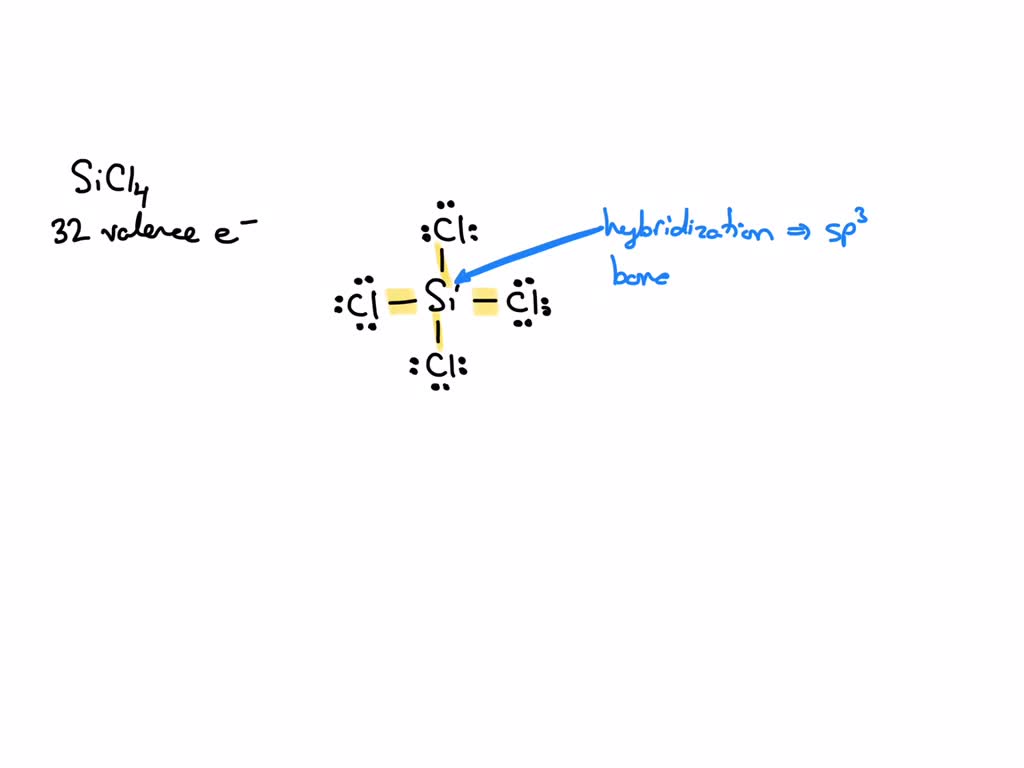
SOLVED: What is the hybridization of the central atom in SiCl4? Hybridization = What are the approximate bond angles in this substance? Bond angles = fill in the blank 2° B. What
sp^3 (sp three) Hybridization (Tetrahedral hybridisation). - Sarthaks eConnect | Largest Online Education Community
Q. Which of the following pair has same hybridisation. 1] BF3 NF3 2] SF4 SiCl4 3] ClO4 ClO2 4] CO2 SiO2

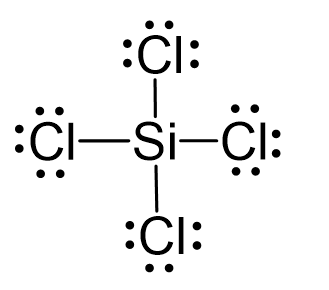
SiCl4 lewis structure, molecular geometry, hybridization, polar or nonpolar | Molecular geometry, Molecular shapes, Molecular
Q. Which of the following pair has same hybridisation. 1] BF3 NF3 2] SF4 SiCl4 3] ClO4 ClO2 4] CO2 SiO2
Silicon tetrachloride SiCl4: Molecular Geometry - Hybridization - Molecular Weight - Molecular Formula - Bond Pairs - Lone Pairs - Lewis structure –

SOLVED: 4a) Using Valence Bond Theory, show the hybridization and bonding scheme for silicon tetrachloride (SiCl4): (a) write the atomic orbital diagram for the central atom, (b) circle the atomic orbitals that
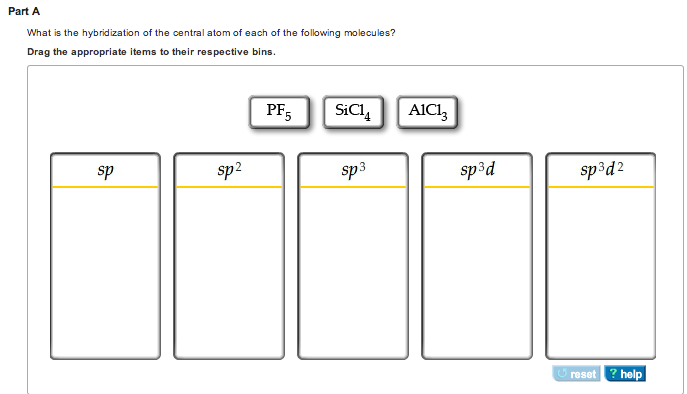
What is the hybridization of the central atom of each of the following molecules? - Home Work Help - Learn CBSE Forum