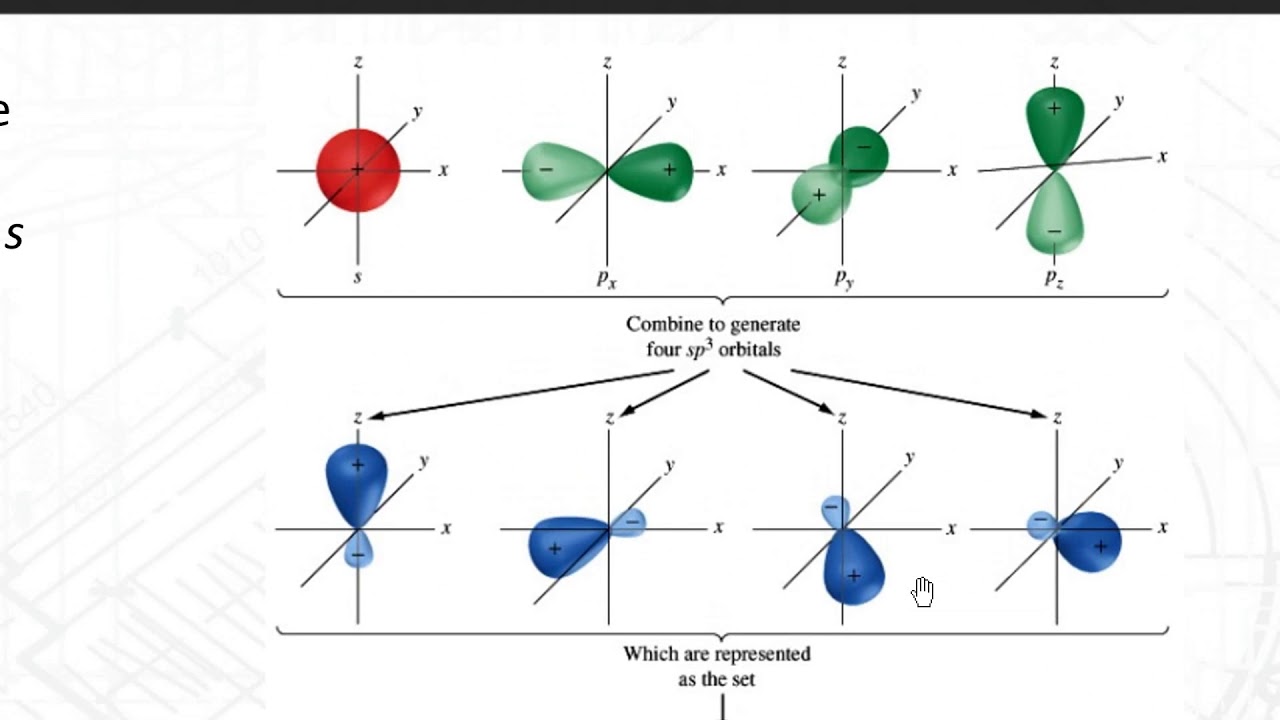
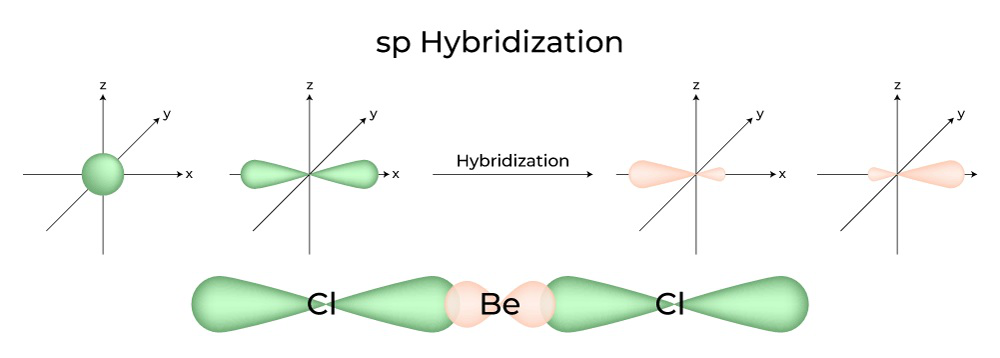
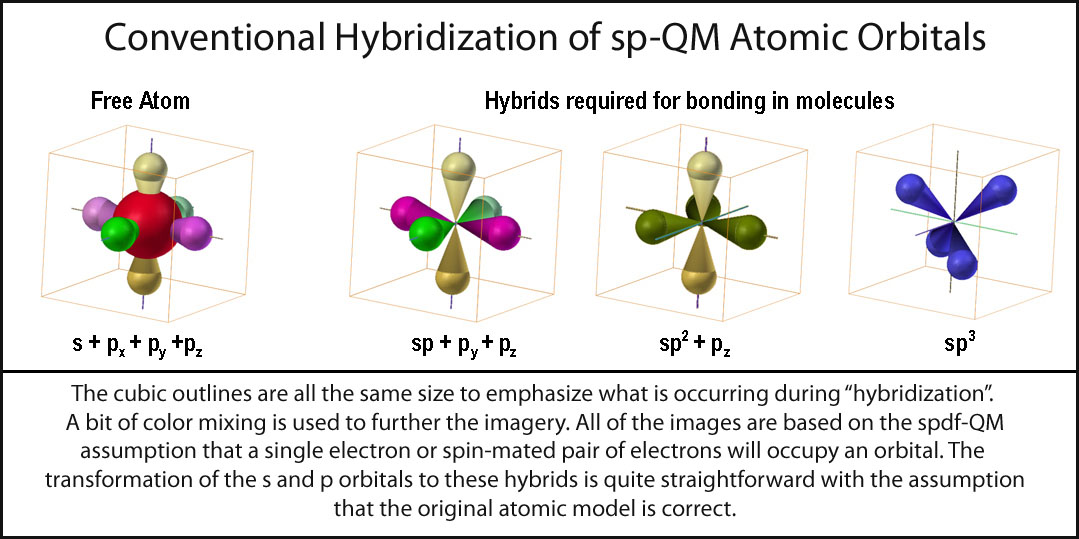
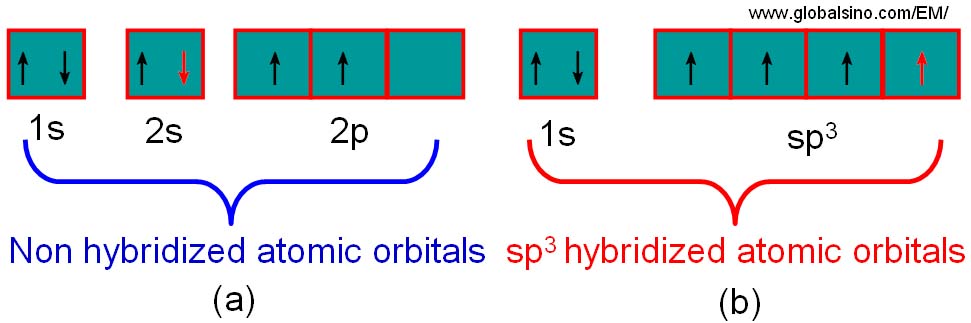
SOLVED: **NOTE: SHOW ALL YOUR WORK AND EXPLANATION. Question 45 5 pts Describe the hybridization of silicon and the molecular structure of the following molecule and ion: (a) (CH3)3SiH (b) SiF2
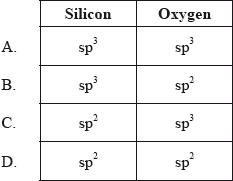
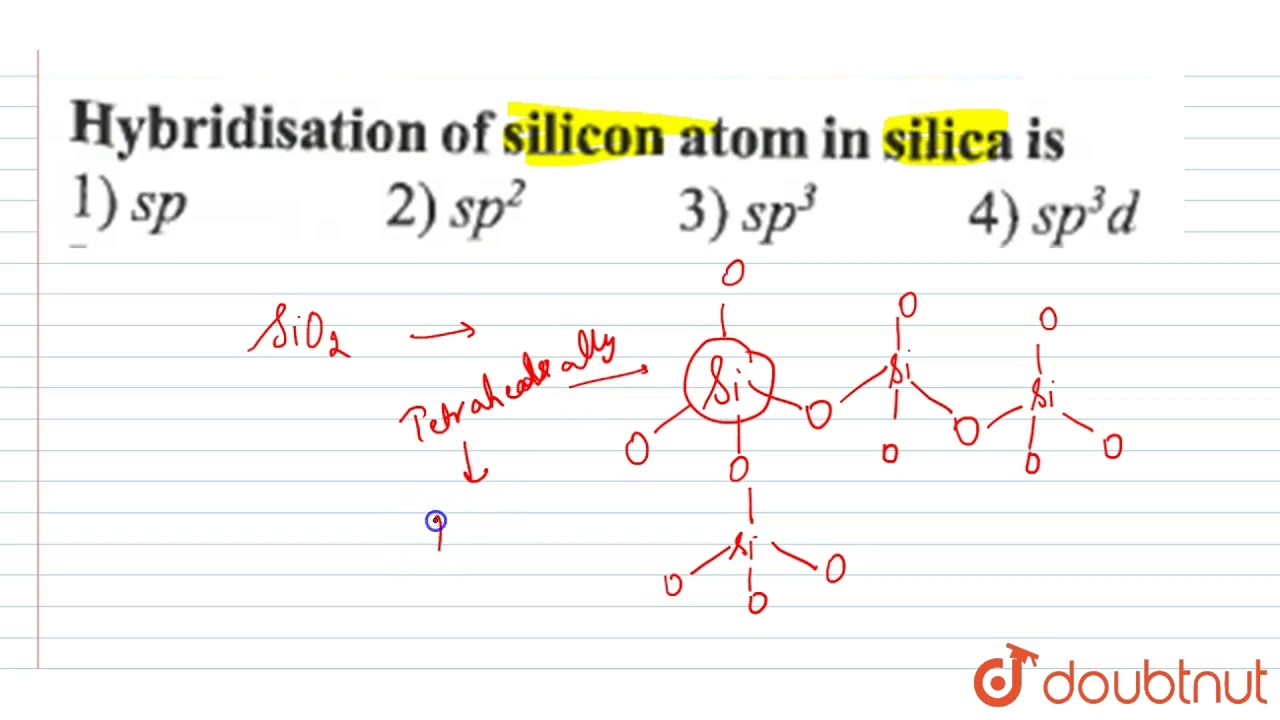
![What is the hybridization of the central atom of $SiO_2$?A.\\[sp\\]B.\\[s{p^2}\\]C.\\[s{p^3}\\]D.\\[s{p^3}d\\] What is the hybridization of the central atom of $SiO_2$?A.\\[sp\\]B.\\[s{p^2}\\]C.\\[s{p^3}\\]D.\\[s{p^3}d\\]](https://www.vedantu.com/question-sets/bfbe5949-20d8-4df4-b2df-ad4de6f9d4df3600507102077243315.png)
What is the hybridization of the central atom of $SiO_2$?A.\\[sp\\]B.\\[s{p^2}\\]C.\\[s{p^3}\\]D.\\[s{p^3}d\\]
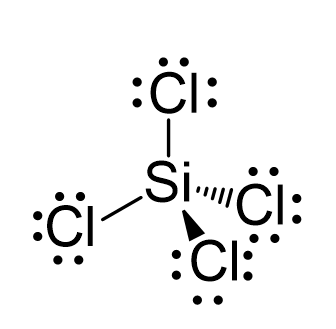
Hybridization is a phenomenon that takes place in an atom before chemical bonding. How is hybridization responsible for the observed structure of SiCl4? - Quora

Silicon tetrafluoride, SiF_4, is a colorless gas formed when hydrofluoric acid attacks silica (SiO_2) or glass. Describe the bonding in the SiF_4 molecule, using valence bond theory. | Homework.Study.com