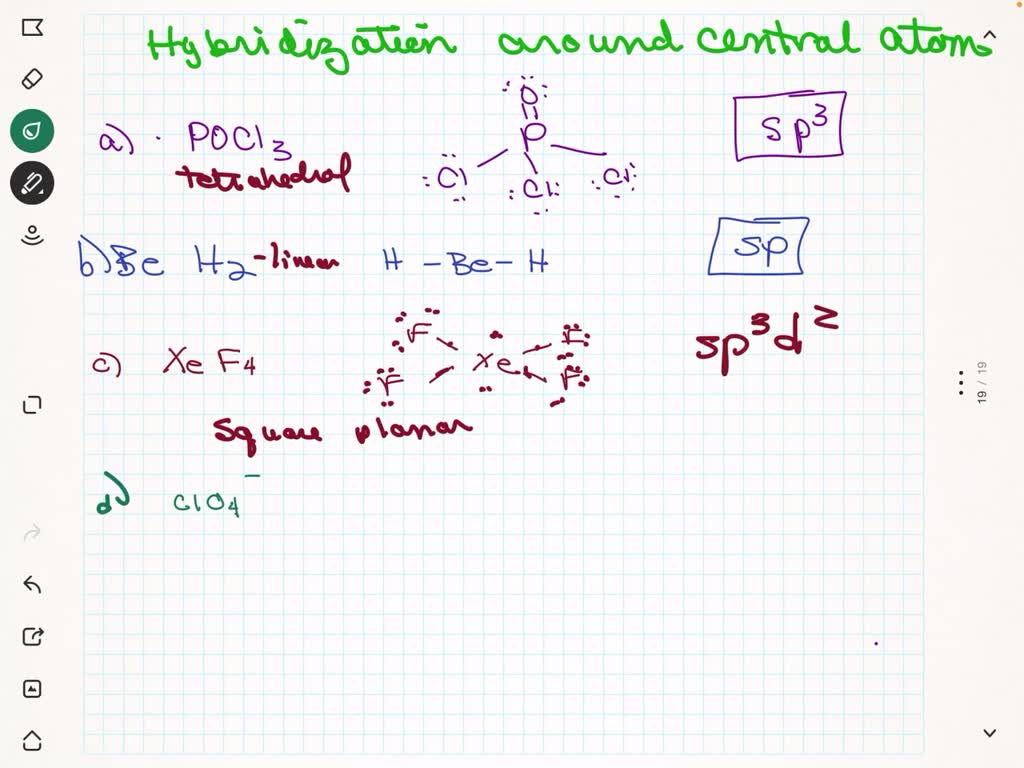
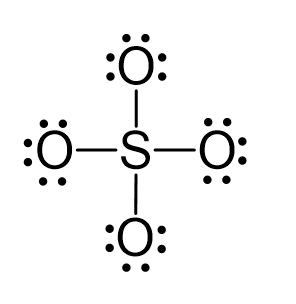
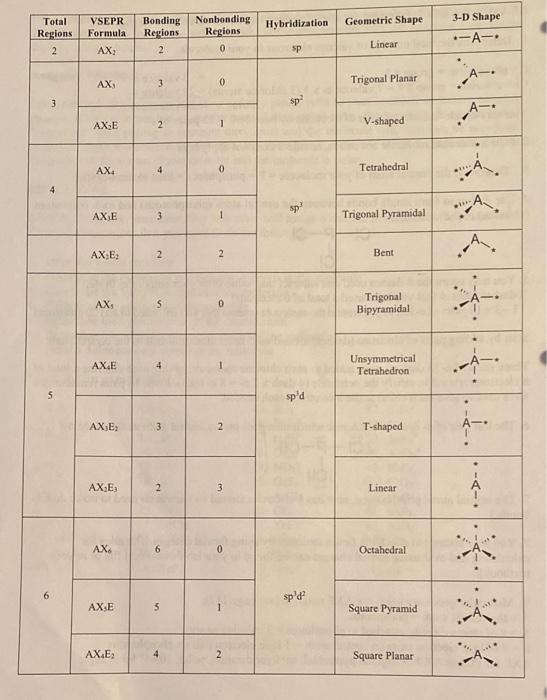
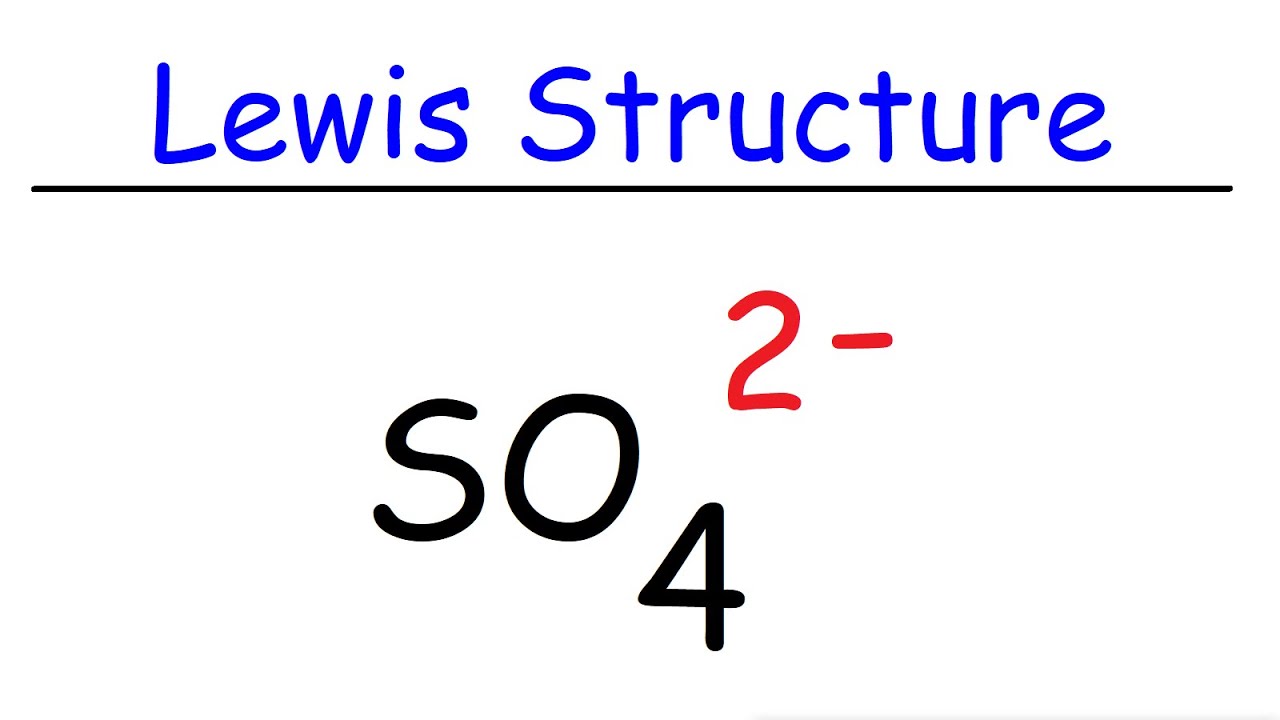
What is the geometric shape of the following molecules, SO4^-2, NCL3, CS2, SO3 using VSEPR theory and what are the hybridization type of those molecules? - Quora
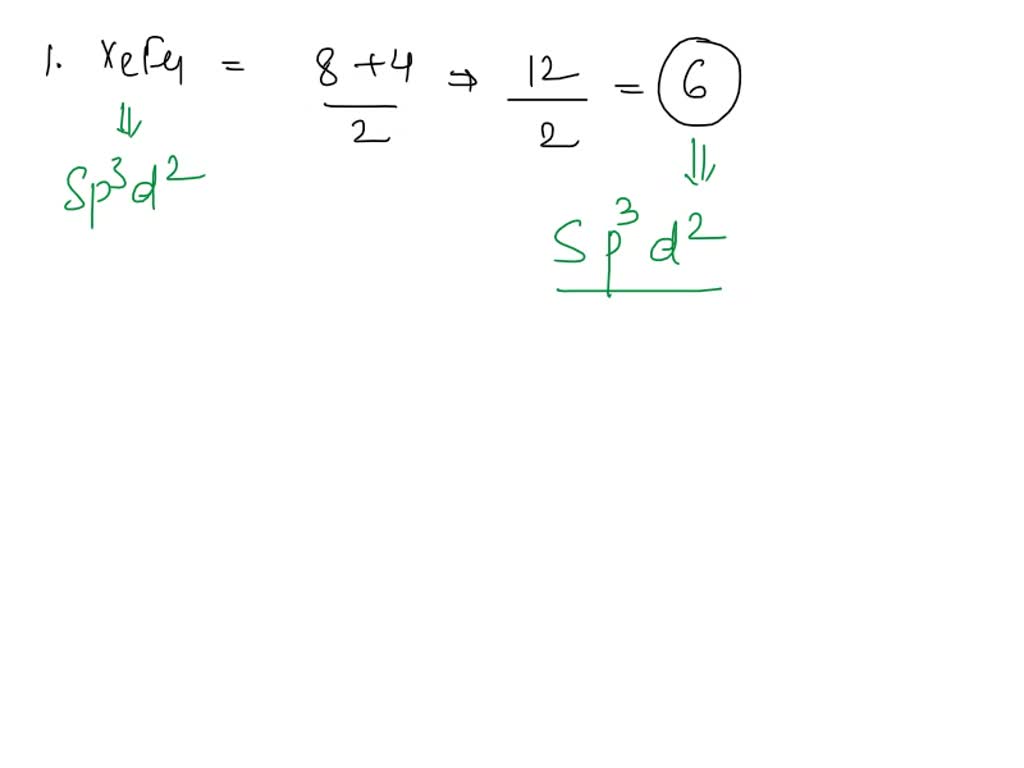
SOLVED: Give the expected hybridization of the central atom for the following molecules or ions: 1. XeF4 2. PH3 3. SO4^2- 4. BH3 5. PO3^3- 6. ClO2- 7. SO4^2-