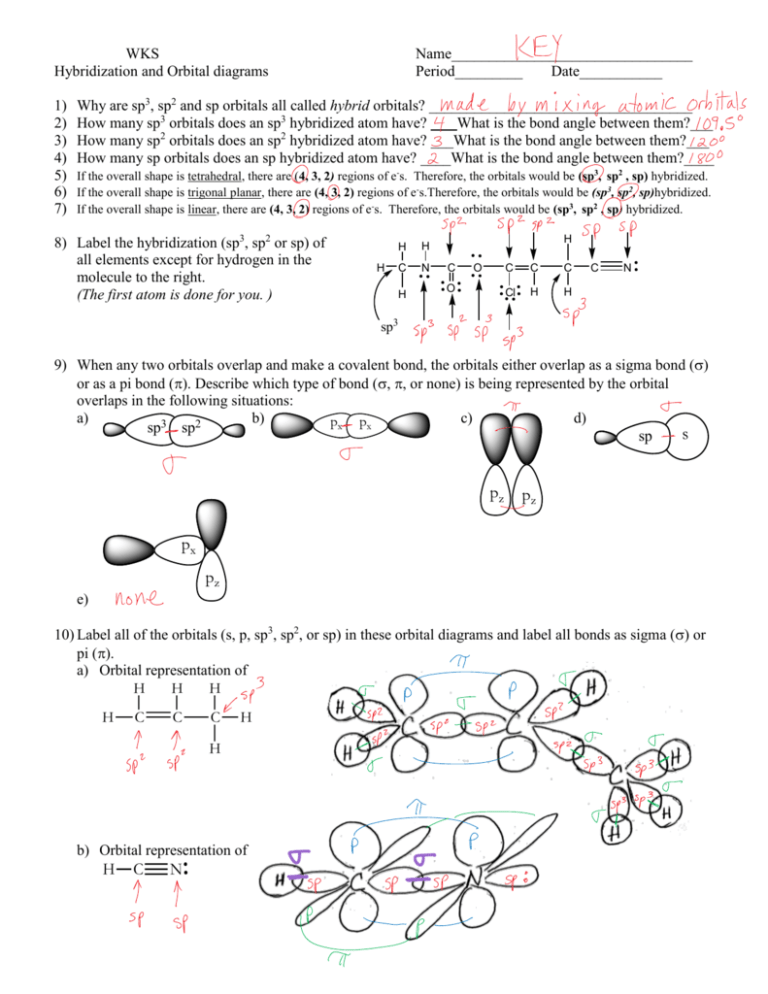
In which of the following molecules does the carbon atom have sp hybridization: HCN, CH4, CO2, and CH2O? - Quora

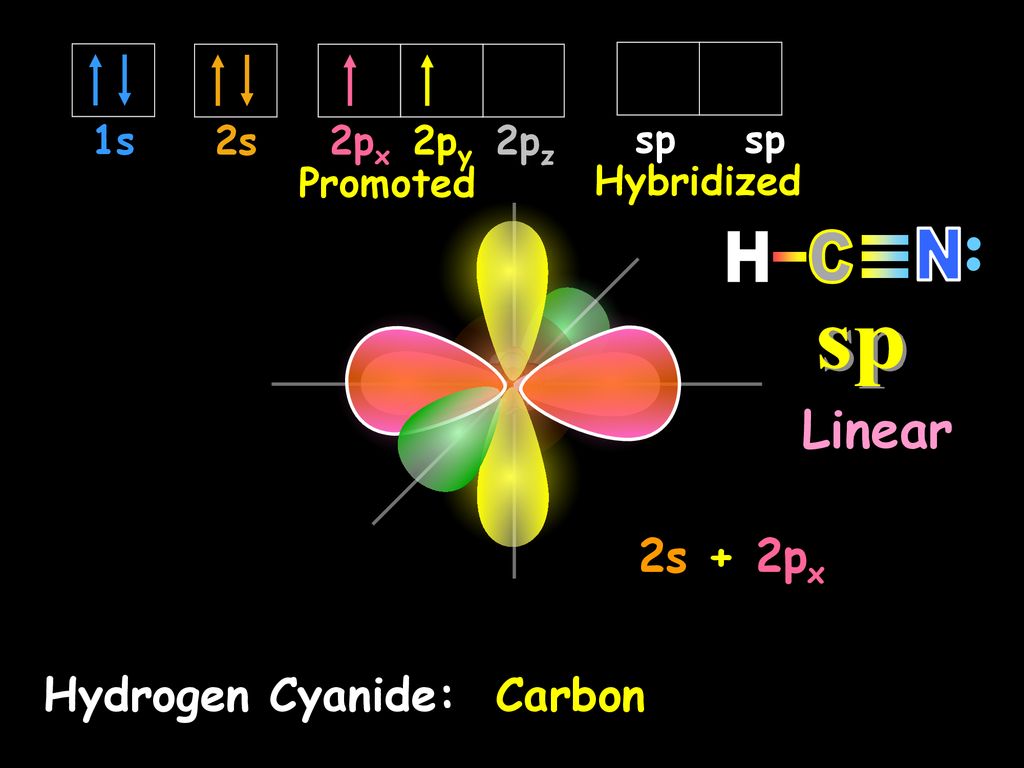
sp hybridization on nitrogen/nitrogen hybridization/HCN hybridization,orbital overlap diagram of hcn - YouTube
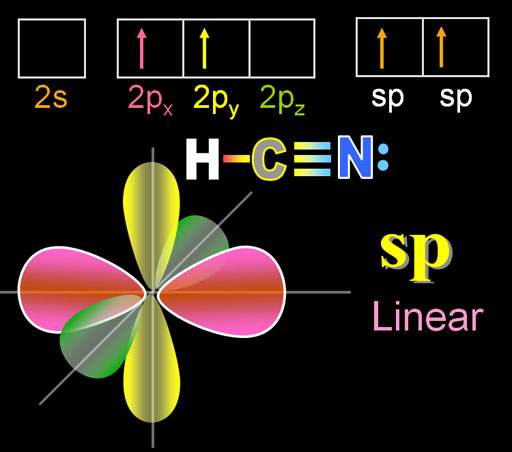
Why does nitrogen in HCN hybridize, instead of forming 3 pi bonds? All 2p orbitals have only 1 electron. - Quora