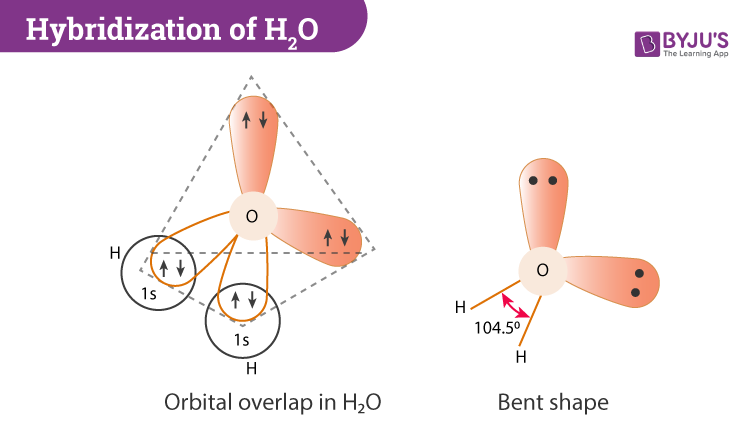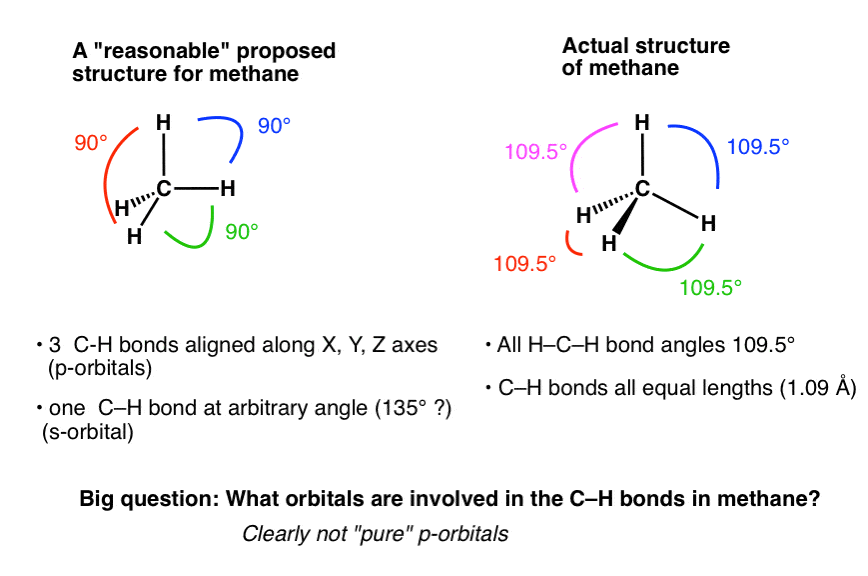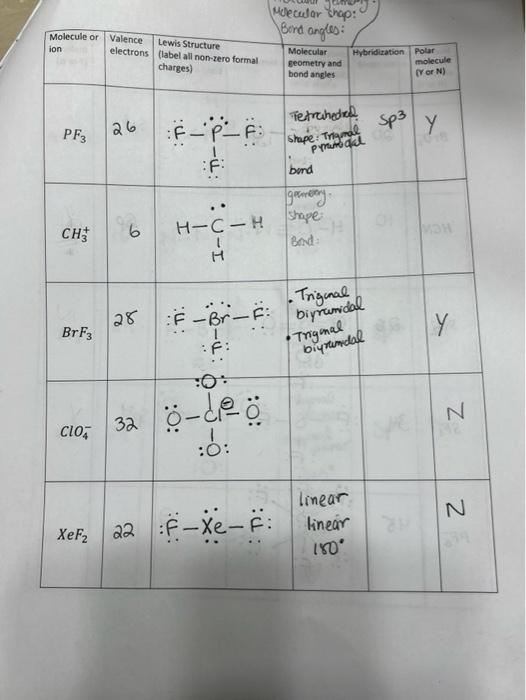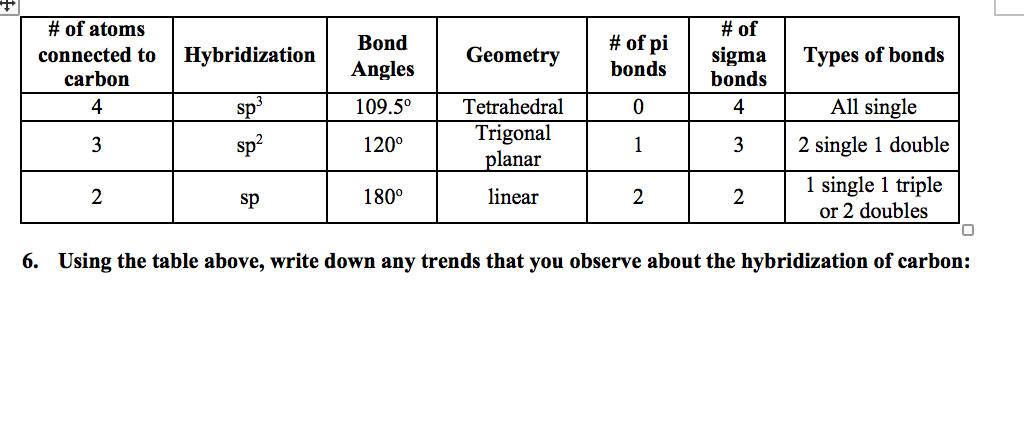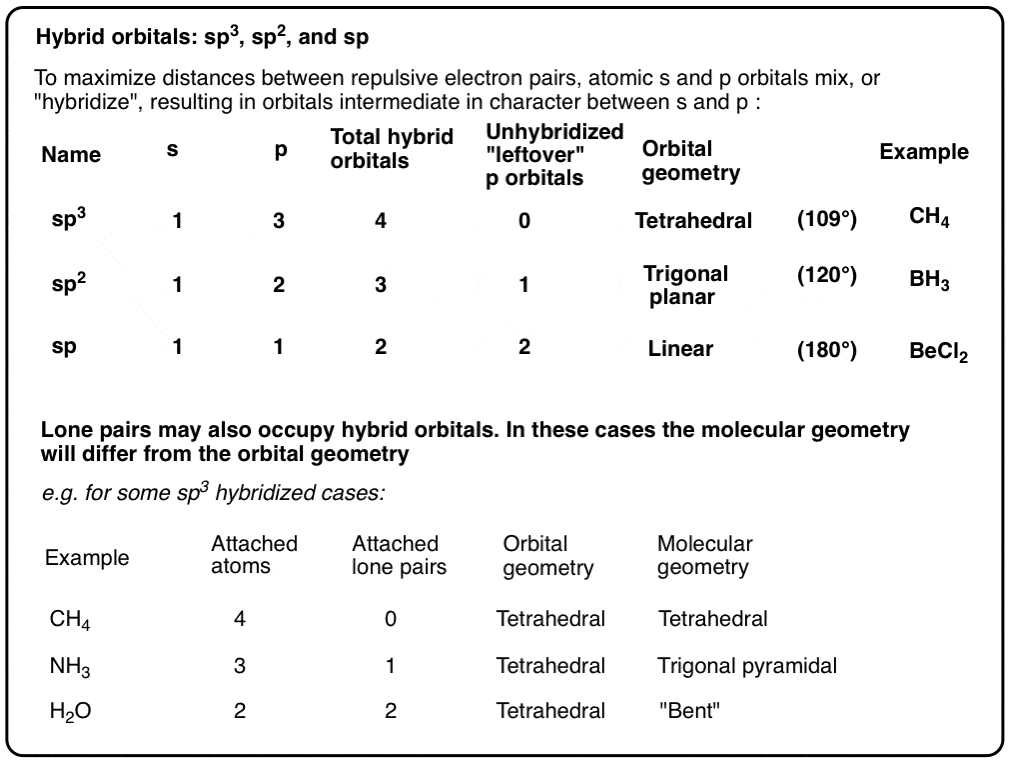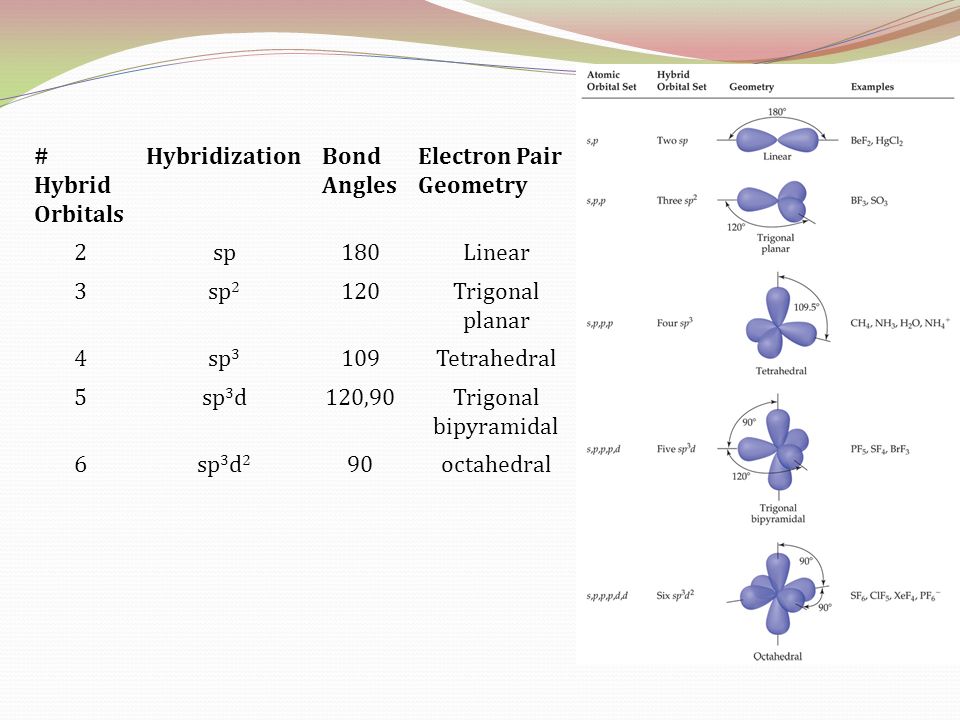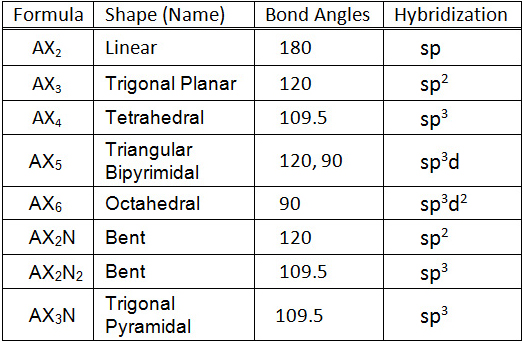
What mode of hybridization is associated with each of the five common electron domain geometries? | Socratic
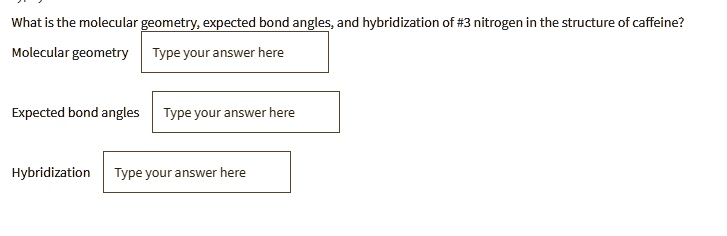
SOLVED: What is the molecular geometry, expected bond angles, and hybridization of nitrogen in the structure of caffeine? Molecular geometry: Type your answer here Expected bond angles: Type your answer here Hybridization:

For each of the following compounds: a) Give the hybridization and approximate bond angles around each atom except hydrogen. b) Draw a three-dimensional diagram, including any lone pairs of electrons. | Homework.Study.com