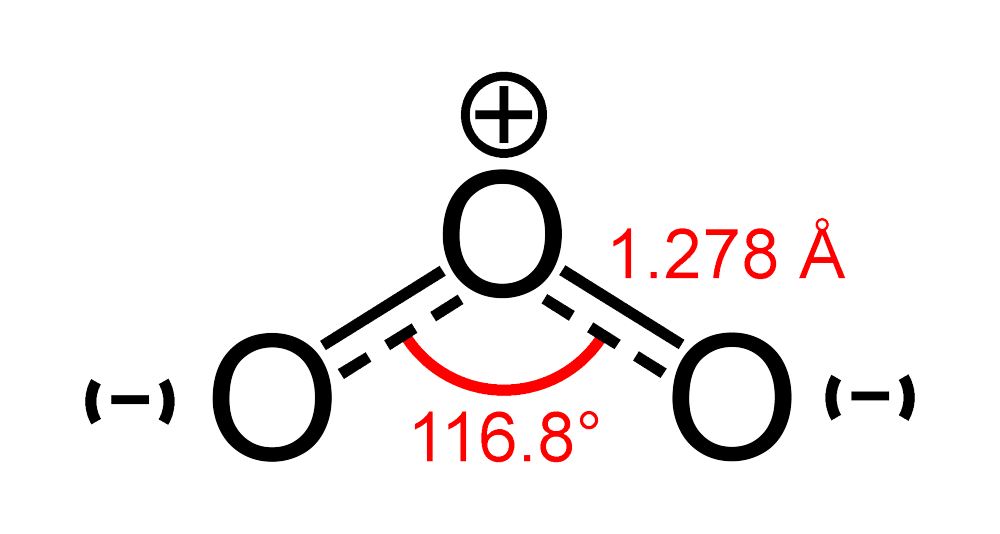
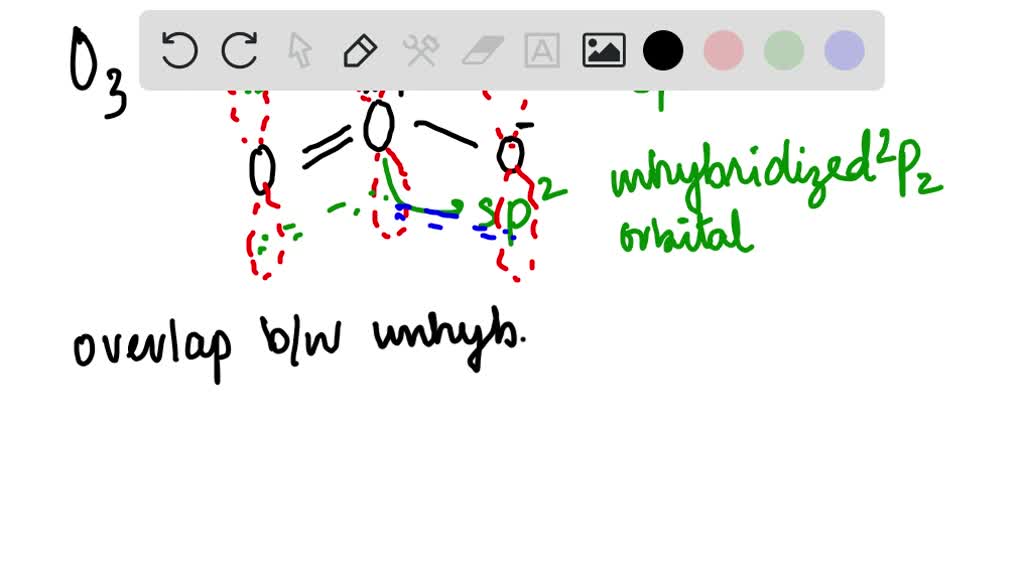
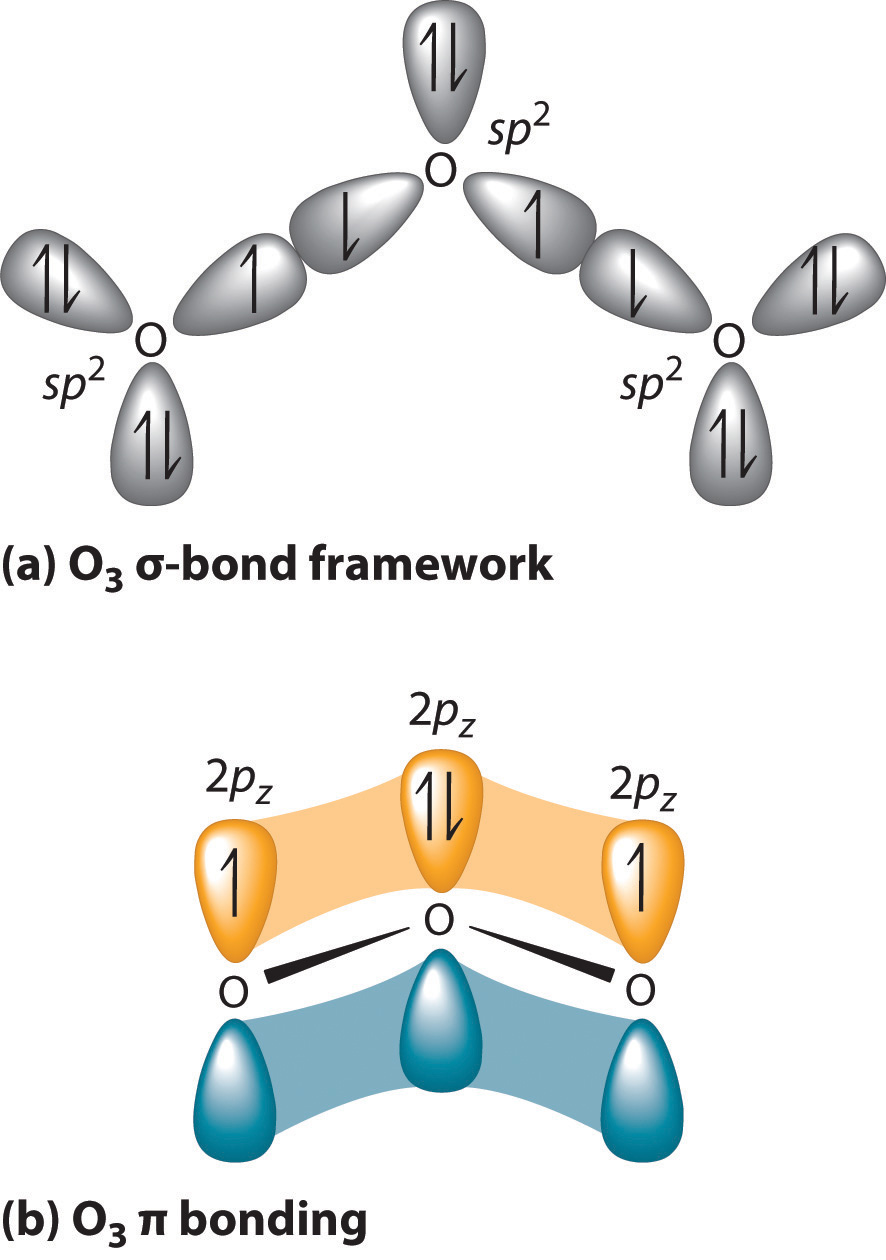
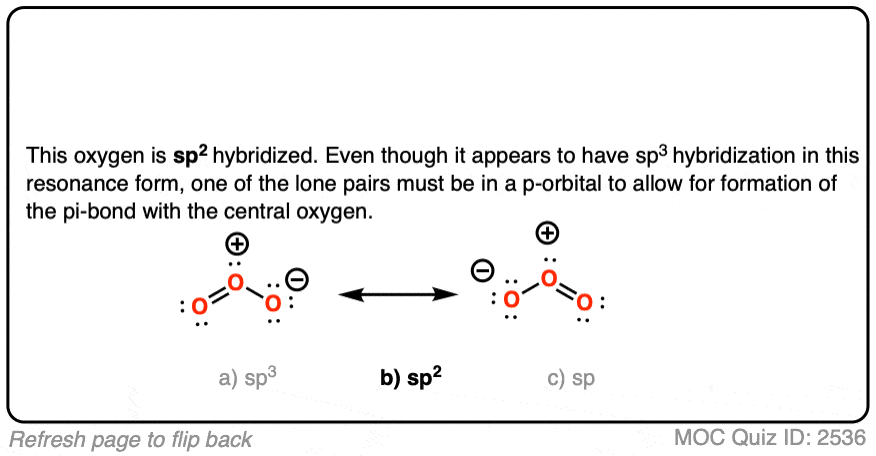
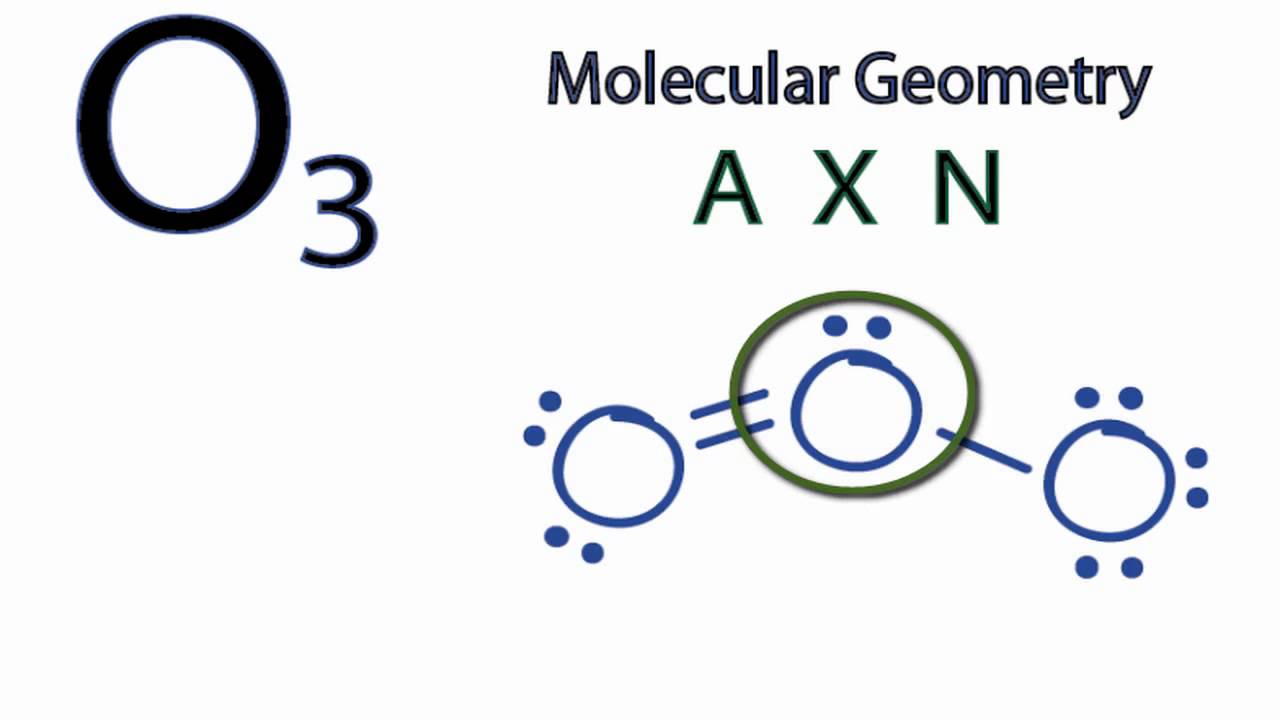
O3 Hybridization (Ozone) | O3 Hybridization (Ozone) O3 is a chemical formula for Ozone molecule. It comprises three Oxygen atoms, out of which one Oxygen atom is in the center... | By

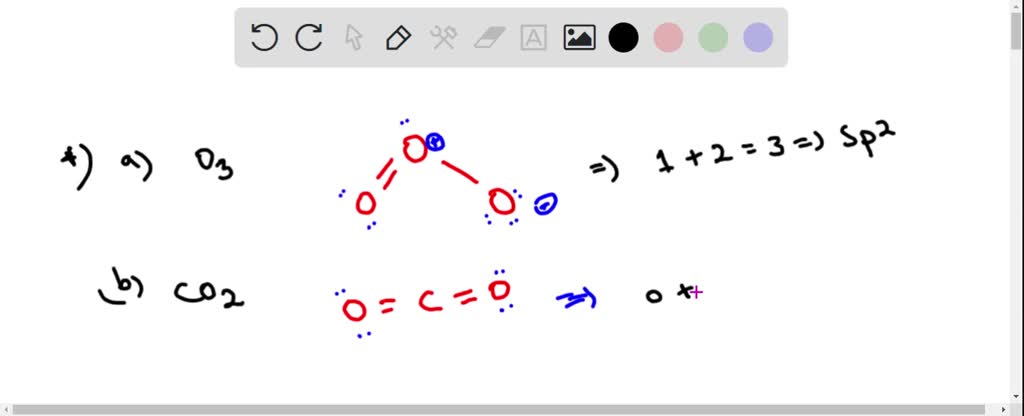
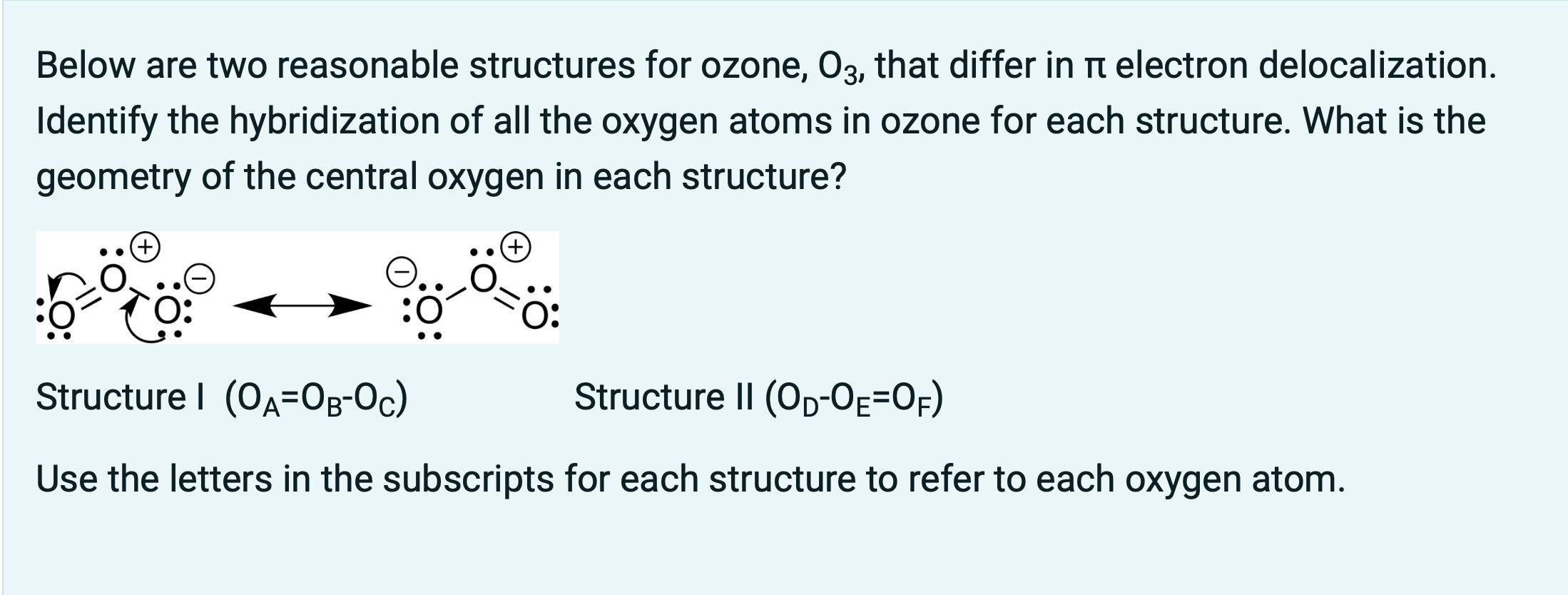
Write Lewis formulas for molecular oxygen and ozone. Assuming that all of the valence electrons in the oxygen atoms are in hybrid orbitals, what would be the hybridization of the oxygen atoms

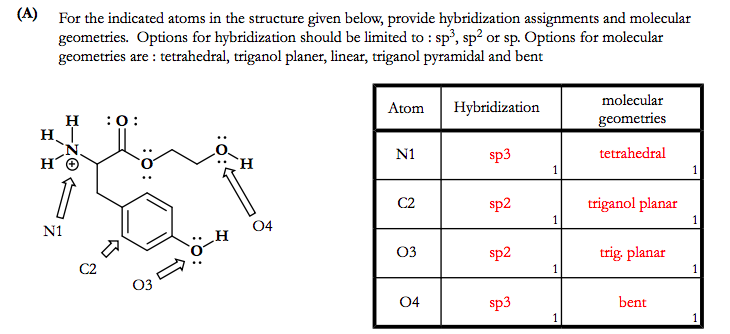
Chapter 10 Bonding and Molecular Structure: Orbital Hybridization and Molecular Orbitals Atoms are bonded together by electrons, but what is a bond? A. - ppt download