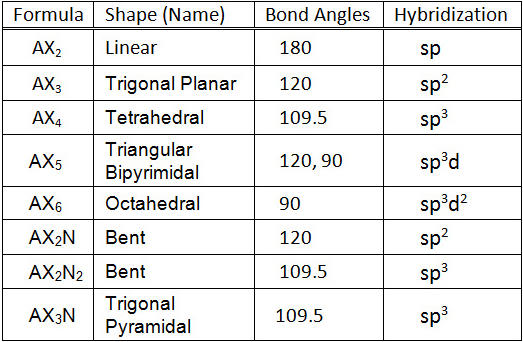
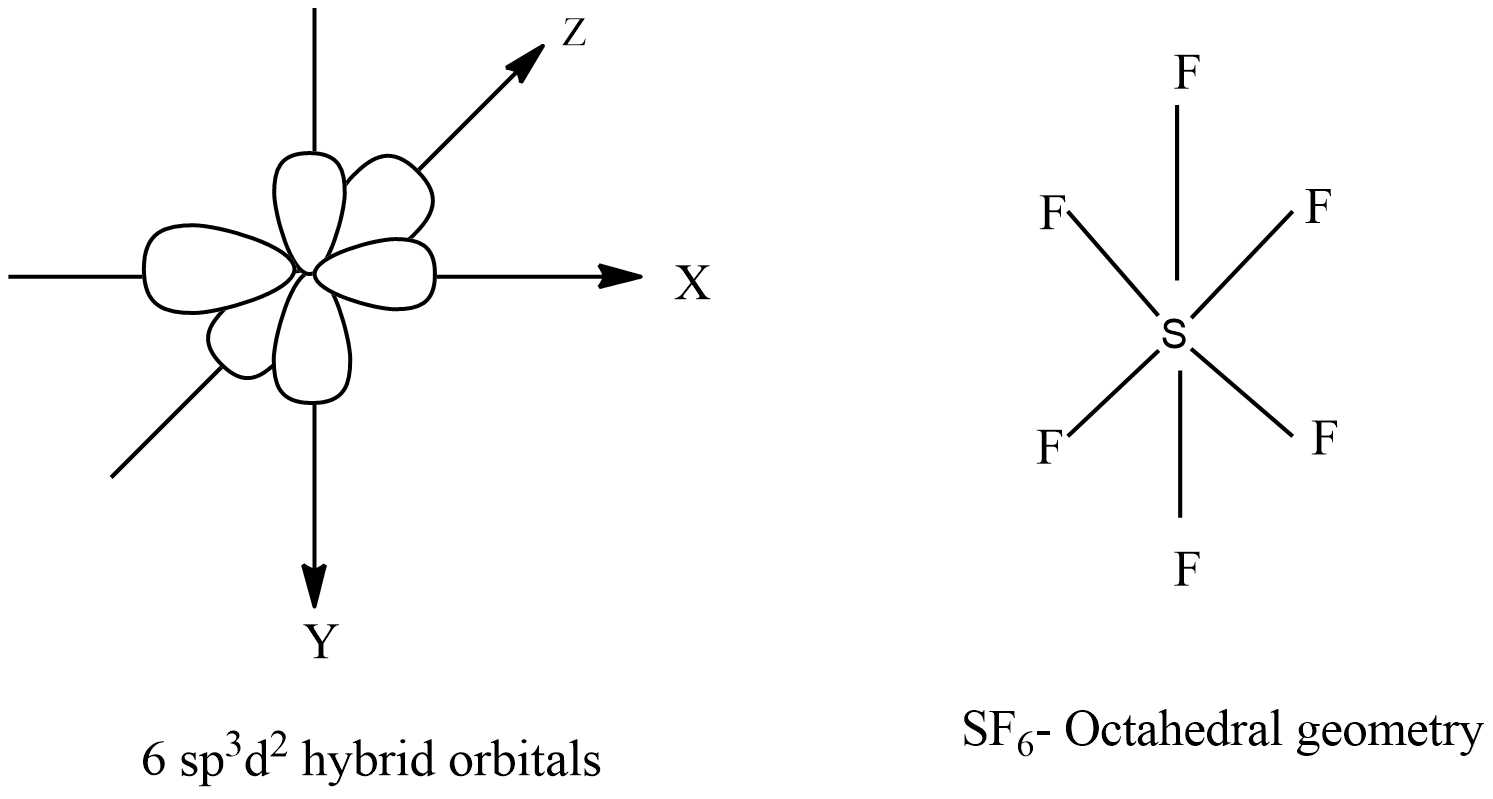
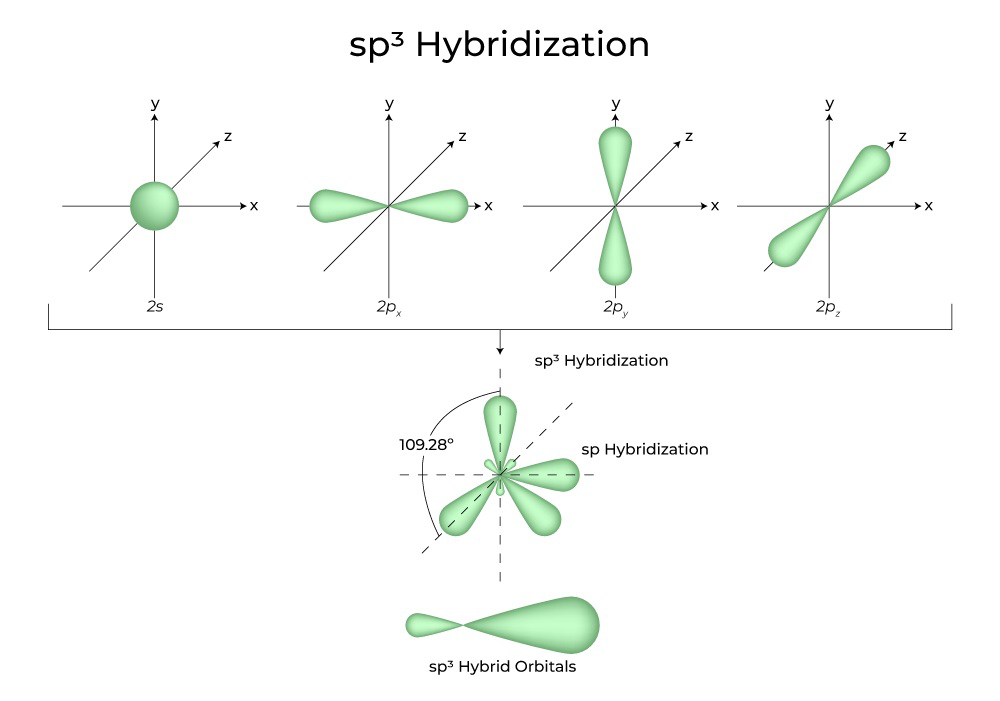
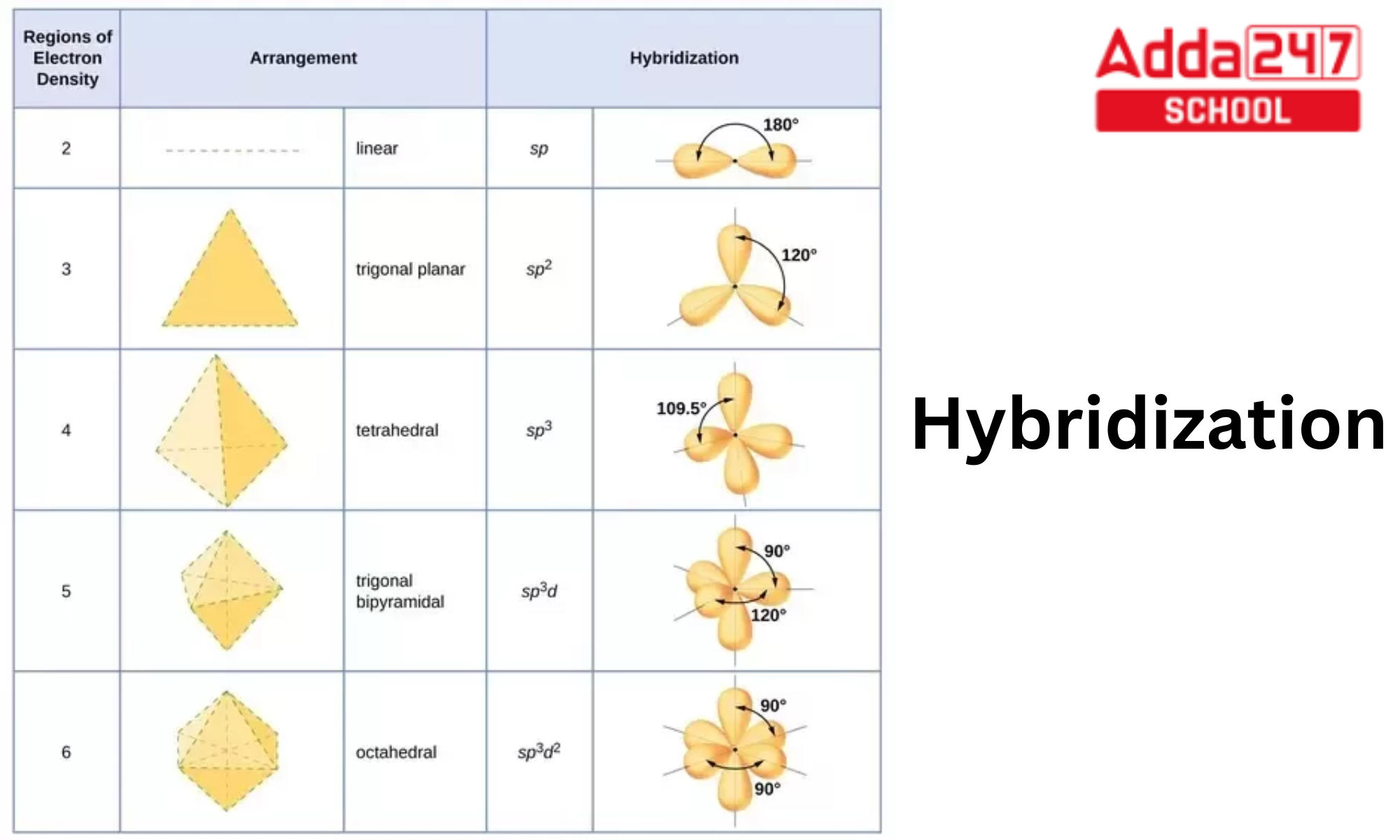
What type(s) of hybridization would you predict for molecules having the following general formula? AB_2U_4 | Homework.Study.com

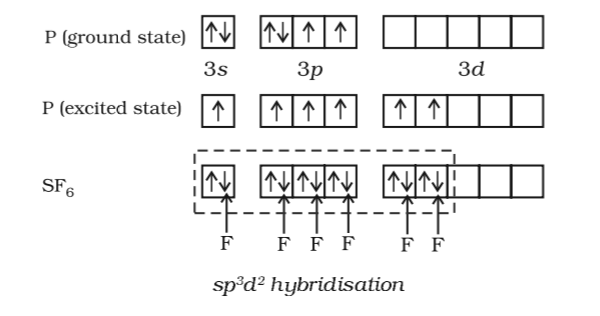
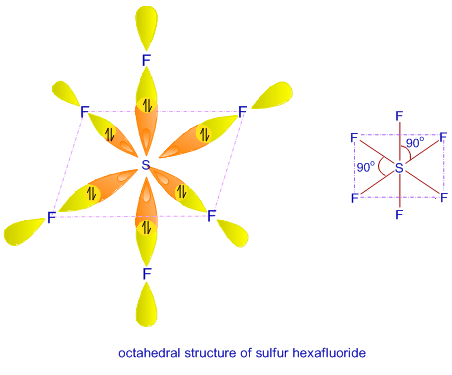
Hybridization || sp3d || sp3d2 || sp3d3 || Formation of PF5, SF6 and IF7 || Chemical Bonding 11th - YouTube

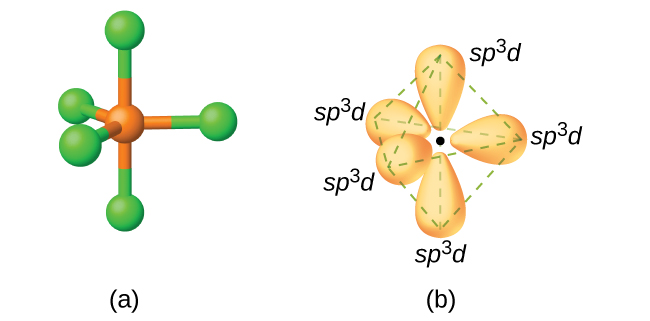
A molecule containing a central atom with sp3d2 hybridization has a(n) ______ electron geometry. a) trigonal planar b) trigonal bipyramidal c) octahedral d) tetrahedral e) trigonal pyramidal | Homework.Study.com
![Which complex compound possesses sp^{3}d^{2} hybridisation?[Fe(NH_{3})_{6}]^{3+}[Fe(CN)_{6}]^{4-}[Fe(CN)_{6}]^{3-}[Fe(Cl)_{6}]^{3-} Which complex compound possesses sp^{3}d^{2} hybridisation?[Fe(NH_{3})_{6}]^{3+}[Fe(CN)_{6}]^{4-}[Fe(CN)_{6}]^{3-}[Fe(Cl)_{6}]^{3-}](https://search-static.byjusweb.com/question-images/toppr_ext/questions/713178_678375_ans_71569d13566849afa3eff10ad47d91bb.png)
Which complex compound possesses sp^{3}d^{2} hybridisation?[Fe(NH_{3})_{6}]^{3+}[Fe(CN)_{6}]^{4-}[Fe(CN)_{6}]^{3-}[Fe(Cl)_{6}]^{3-}
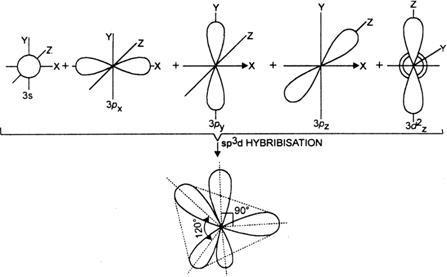
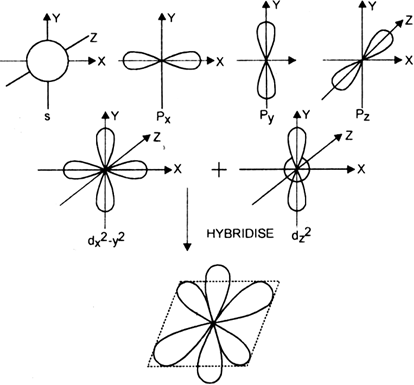
State and explain the geometric arrangements possible in sp3d and sp3d2 hybridisation. Name the d-orbitals involved in these. - Zigya